Oncostatin M: From Intracellular Signaling to Therapeutic Targets in Liver Cancer
- PMID: 36077744
- PMCID: PMC9454586
- DOI: 10.3390/cancers14174211
Oncostatin M: From Intracellular Signaling to Therapeutic Targets in Liver Cancer
Abstract
Primary liver cancers represent the third-most-common cause of cancer-related mortality worldwide, with an incidence of 80-90% for hepatocellular carcinoma (HCC) and 10-15% for cholangiocarcinoma (CCA), and an increasing morbidity and mortality rate. Although HCC and CCA originate from independent cell populations (hepatocytes and biliary epithelial cells, respectively), they develop in chronically inflamed livers. Evidence obtained in the last decade has revealed a role for cytokines of the IL-6 family in the development of primary liver cancers. These cytokines operate through the receptor subunit gp130 and the downstream Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling pathways. Oncostatin M (OSM), a member of the IL-6 family, plays a significant role in inflammation, autoimmunity, and cancer, including liver tumors. Although, in recent years, therapeutic approaches for the treatment of HCC and CCA have been implemented, limited treatment options with marginal clinical benefits are available. We discuss how OSM-related pathways can be selectively inhibited and therapeutically exploited for the treatment of liver malignancies.
Keywords: cholangiocarcinoma; epithelial–mesenchymal transition; hepatocellular carcinoma; inflammation; signaling; tumor microenvironment; tumor proliferation.
Conflict of interest statement
The authors declare no conflict of interest.
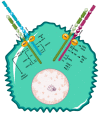
Figures

References
-
- Calderaro J., Couchy G., Imbeaud S., Amaddeo G., Letouzé E., Blanc J.F., Laurent C., Hajji Y., Azoulay D., Bioulac-Sage P., et al. Histological subtypes of hepatocellular carcinoma are related to gene mutations and molecular tumour classification. J. Hepatol. 2017;67:727–738. doi: 10.1016/j.jhep.2017.05.014. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

