Salivary Gland Adaptation to Dietary Inclusion of Hydrolysable Tannins in Boars
- PMID: 36077892
- PMCID: PMC9454789
- DOI: 10.3390/ani12172171
Salivary Gland Adaptation to Dietary Inclusion of Hydrolysable Tannins in Boars
Abstract
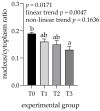
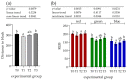
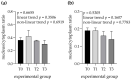
The ingestion of hydrolysable tannins as a potential nutrient to reduce boar odor in entire males results in the significant enlargement of parotid glands (parotidomegaly). The objective of this study was to characterize the effects of different levels of hydrolysable tannins in the diet of fattening boars (n = 24) on salivary gland morphology and proline-rich protein (PRP) expression at the histological level. Four treatment groups of pigs (n = 6 per group) were fed either a control (T0) or experimental diet, where the T0 diet was supplemented with 1% (T1), 2% (T2), or 3% (T3) of the hydrolysable tannin-rich extract Farmatan®. After slaughter, the parotid and mandibular glands of the experimental pigs were harvested and dissected for staining using Goldner's Trichrome method, and immunohistochemical studies with antibodies against PRPs. Morphometric analysis was performed on microtome sections of both salivary glands, to measure the acinar area, the lobular area, the area of the secretory ductal cells, and the sizes of glandular cells and their nuclei. Histological assessment revealed that significant parotidomegaly was only present in the T3 group, based on the presence of larger glandular lobules, acinar areas, and their higher nucleus to cytoplasm ratio. The immunohistochemical method, supported by color intensity measurements, indicated significant increases in basic PRPs (PRB2) in the T3 and acidic PRPs (PRH1/2) in the T1 groups. Tannin supplementation did not affect the histo-morphological properties of the mandibular gland. This study confirms that pigs can adapt to a tannin-rich diet by making structural changes in their parotid salivary gland, indicating its higher functional activity.
Keywords: dietary supplements; histology; immunohistochemistry; mandibular gland; parotid gland; pigs; proline-rich proteins (PRP); tannins.
Conflict of interest statement
The authors declare that there are no conflict of interest.
Figures





References
-
- Cappai M.G., Wolf P., Pinna W., Kamphues J. Dose-Response Relationship between Dietary Polyphenols from Acorns and Parotid Gland Hypertrophy in Pigs. Food Nutr. Sci. 2012;3:1261–1268. doi: 10.4236/fns.2012.39166. - DOI
-
- Mueller-Harvey I. Unravelling the conundrum of tannins in animal nutrition and health. J. Sci. Food Agric. 2006;86:2010–2037. doi: 10.1002/jsfa.2577. - DOI
-
- Flis M., Sobotka W., Antoszkiewicz Z., Lipiński K., Zduńczyk Z. The effect of grain polyphenols and the addition of vitamin E to diets enriched with α-linolenic acid on the antioxidant status of pigs. J. Anim. Feed Sci. 2010;19:539–553. doi: 10.22358/jafs/66319/2010. - DOI
-
- Lipiński K., Antoszkiewicz Z., Mazur-Kuśnirek M., Korniewicz D., Kotlarczyk S. The effect of polyphenols on the performance and antioxidant status of sows and piglets. Ital. J. Anim. Sci. 2019;18:174–181. doi: 10.1080/1828051X.2018.1503043. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

