Detailed Investigation of Factors Affecting the Synthesis of SiO2@Au for the Enhancement of Raman Spectroscopy
- PMID: 36080115
- PMCID: PMC9458010
- DOI: 10.3390/nano12173080
Detailed Investigation of Factors Affecting the Synthesis of SiO2@Au for the Enhancement of Raman Spectroscopy
Abstract
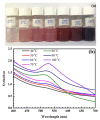
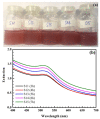
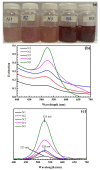
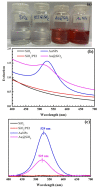
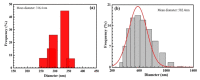
The reaction time, temperature, ratio of precursors, and concentration of sodium citrate are known as the main factors that affect the direct synthesis process of SiO2@Au based on the chemical reaction of HAuCl4 and sodium citrate. Hence, we investigated, in detail, and observed that these factors played a crucial role in determining the shape and size of synthesized nanoparticles. The significant enhancement of the SERS signal corresponding to the fabrication conditions is an existing challenge. Our study results show that the optimal reaction conditions for the fabrication of SiO2@Au are a 1:21 ratio of HAuCl4 to sodium citrate, with an initial concentration of sodium citrate of 4.2 mM, and a reaction time lasting longer than 6 h at a temperature of 80 °C. Under optimal conditions, our synthesis process result is SiO2@Au nanoparticles with a diameter of approximately 350 nm. In particular, the considerable enhancement of Raman intensities of SiO2@Au compared to SiO2 particles was examined.
Keywords: Raman spectroscopy; SERS; SiO2@Au; core shell.
Conflict of interest statement
The authors declare no conflict of interest.
Figures








References
-
- Peeters H., Keulemans M., Nuyts G., Vanmeert F., Li C., Minjauw M., Detavernier C., Bals S., Lenaerts S., Verbruggen S.W. Plasmonic gold-embedded TiO2 thin films as photocatalytic self-cleaning coatings. Appl. Catal. B Environ. 2020;267:118654. doi: 10.1016/j.apcatb.2020.118654. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

