Multifaceted roles of SARM1 in axon degeneration and signaling
- PMID: 36090788
- PMCID: PMC9453223
- DOI: 10.3389/fncel.2022.958900
Multifaceted roles of SARM1 in axon degeneration and signaling
Abstract
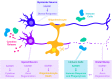
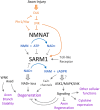
Axons are considered to be particularly vulnerable components of the nervous system; impairments to a neuron's axon leads to an effective silencing of a neuron's ability to communicate with other cells. Nervous systems have therefore evolved plasticity mechanisms for adapting to axonal damage. These include acute mechanisms that promote the degeneration and clearance of damaged axons and, in some cases, the initiation of new axonal growth and synapse formation to rebuild lost connections. Here we review how these diverse processes are influenced by the therapeutically targetable enzyme SARM1. SARM1 catalyzes the breakdown of NAD+, which, when unmitigated, can lead to rundown of this essential metabolite and axonal degeneration. SARM1's enzymatic activity also triggers the activation of downstream signaling pathways, which manifest numerous functions for SARM1 in development, innate immunity and responses to injury. Here we will consider the multiple intersections between SARM1 and the injury signaling pathways that coordinate cellular adaptations to nervous system damage.
Keywords: MAP Kinase; Nicotinamide Adenine Dinucleotide (NAD); SARM1; TIR domain; axon degeneration; axonal transport; injury signaling.
Copyright © 2022 Waller and Collins.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
SARM1 activation triggers axon degeneration locally via NAD⁺ destruction.Science. 2015 Apr 24;348(6233):453-7. doi: 10.1126/science.1258366. Epub 2015 Apr 23. Science. 2015. PMID: 25908823 Free PMC article.
-
SARM1-specific motifs in the TIR domain enable NAD+ loss and regulate injury-induced SARM1 activation.Proc Natl Acad Sci U S A. 2016 Oct 11;113(41):E6271-E6280. doi: 10.1073/pnas.1601506113. Epub 2016 Sep 26. Proc Natl Acad Sci U S A. 2016. PMID: 27671644 Free PMC article.
-
The axon degeneration gene SARM1 is evolutionarily distinct from other TIR domain-containing proteins.Mol Genet Genomics. 2017 Aug;292(4):909-922. doi: 10.1007/s00438-017-1320-6. Epub 2017 Apr 26. Mol Genet Genomics. 2017. PMID: 28447196
-
NAD+, Axonal Maintenance, and Neurological Disease.Antioxid Redox Signal. 2023 Dec;39(16-18):1167-1184. doi: 10.1089/ars.2023.0350. Epub 2023 Sep 7. Antioxid Redox Signal. 2023. PMID: 37503611 Free PMC article. Review.
-
Mechanism of initiation and regulation of axonal degeneration with special reference to NMNATs and Sarm1.Neurosci Res. 2023 Dec;197:3-8. doi: 10.1016/j.neures.2021.11.002. Epub 2021 Nov 9. Neurosci Res. 2023. PMID: 34767875 Review.
Cited by
-
High-Resolution Proteomics Unravel a Native Functional Complex of Cav1.3, SK3, and Hyperpolarization-Activated Cyclic Nucleotide-Gated Channels in Midbrain Dopaminergic Neurons.Cells. 2024 May 30;13(11):944. doi: 10.3390/cells13110944. Cells. 2024. PMID: 38891076 Free PMC article.
-
SARM1 Inhibition Maintains Axonal Integrity After Rat Sciatic Nerve Transection and Repair.J Hand Surg Am. 2025 Jan 29:S0363-5023(24)00617-8. doi: 10.1016/j.jhsa.2024.12.009. Online ahead of print. J Hand Surg Am. 2025. PMID: 39895440 Free PMC article.
-
SARM1: a key multifaceted component in immunoregulation, inflammation and neurodegeneration.Front Immunol. 2025 May 13;16:1521364. doi: 10.3389/fimmu.2025.1521364. eCollection 2025. Front Immunol. 2025. PMID: 40433385 Free PMC article. Review.
-
Axonal degeneration in chemotherapy-induced peripheral neurotoxicity: clinical and experimental evidence.J Neurol Neurosurg Psychiatry. 2023 Nov;94(11):962-972. doi: 10.1136/jnnp-2021-328323. Epub 2023 Apr 4. J Neurol Neurosurg Psychiatry. 2023. PMID: 37015772 Free PMC article. Review.
-
Abnormal Intraepidermal Nerve Fiber Density in Disease: A Scoping Review.medRxiv [Preprint]. 2023 Feb 8:2023.02.08.23285644. doi: 10.1101/2023.02.08.23285644. medRxiv. 2023. Update in: Front Neurol. 2023 Apr 20;14:1161077. doi: 10.3389/fneur.2023.1161077. PMID: 36798392 Free PMC article. Updated. Preprint.
References
-
- Angeletti C., Amici A., Gilley J., Loreto A., Trapanotto A. G., Antoniou C., et al. (2022). SARM1 is a multi-functional NAD(P)ase with prominent base exchange activity, all regulated by multiple physiologically relevant NAD metabolites. iScience 25:103812. 10.1016/j.isci.2022.103812 - DOI - PMC - PubMed
-
- Bisby M. A., Chen S. (1990). Delayed wallerian degeneration in sciatic nerves of C57BL/Ola mice is associated with impaired regeneration of sensory axons. Brain Res. 530 117–120. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

