Effects of HSD11B1 knockout and overexpression on local cortisol production and differentiation of mesenchymal stem cells
- PMID: 36091434
- PMCID: PMC9453430
- DOI: 10.3389/fbioe.2022.953034
Effects of HSD11B1 knockout and overexpression on local cortisol production and differentiation of mesenchymal stem cells
Abstract
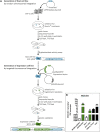
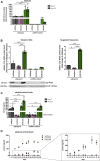
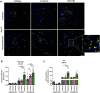
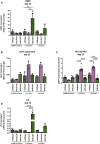
Exogenous glucocorticoids increase the risk for osteoporosis, but the role of endogenous glucocorticoids remains elusive. Here, we describe the generation and validation of a loss- and a gain-of-function model of the cortisol producing enzyme 11β-HSD1 (HSD11B1) to modulate the endogenous glucocorticoid conversion in SCP-1 cells - a model for human mesenchymal stem cells capable of adipogenic and osteogenic differentiation. CRISPR-Cas9 was successfully used to generate a cell line carrying a single base duplication and a 5 bp deletion in exon 5, leading to missense amino acid sequences after codon 146. These inactivating genomic alterations were validated by deep sequencing and by cloning with subsequent capillary sequencing. 11β-HSD1 protein levels were reduced by 70% in the knockout cells and cortisol production was not detectable. Targeted chromosomal integration was used to stably overexpress HSD11B1. Compared to wildtype cells, HSD11B1 overexpression resulted in a 7.9-fold increase in HSD11B1 mRNA expression, a 5-fold increase in 11β-HSD1 protein expression and 3.3-fold increase in extracellular cortisol levels under adipogenic differentiation. The generated cells were used to address the effects of 11β-HSD1 expression on adipogenic and osteogenic differentiation. Compared to the wildtype, HSD11B1 overexpression led to a 3.7-fold increase in mRNA expression of lipoprotein lipase (LPL) and 2.5-fold increase in lipid production under adipogenic differentiation. Under osteogenic differentiation, HSD11B1 knockout led to enhanced alkaline phosphatase (ALP) activity and mRNA expression, and HSD11B1 overexpression resulted in a 4.6-fold and 11.7-fold increase in mRNA expression of Dickkopf-related protein 1 (DKK1) and LPL, respectively. Here we describe a HSD11B1 loss- and gain-of-function model in SCP-1 cells at genetic, molecular and functional levels. We used these models to study the effects of endogenous cortisol production on mesenchymal stem cell differentiation and demonstrate an 11β-HSD1 dependent switch from osteogenic to adipogenic differentiation. These results might help to better understand the role of endogenous cortisol production in osteoporosis on a molecular and cellular level.
Keywords: 11β-HSD1; CRISPR-Cas9; glucocorticoids; osteoporosis; targeted chromosomal integration.
Copyright © 2022 Kragl, Schoon, Tzvetkova, Wenzel, Blaschke, Böcker, Siggelkow and Tzvetkov.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
The rise in expression and activity of 11β-HSD1 in human mesenchymal progenitor cells induces adipogenesis through increased local cortisol synthesis.J Steroid Biochem Mol Biol. 2021 Jun;210:105850. doi: 10.1016/j.jsbmb.2021.105850. Epub 2021 Feb 24. J Steroid Biochem Mol Biol. 2021. PMID: 33639236
-
Osteoblastic 11beta-hydroxysteroid dehydrogenase type 1 activity increases with age and glucocorticoid exposure.J Bone Miner Res. 2002 Jun;17(6):979-86. doi: 10.1359/jbmr.2002.17.6.979. J Bone Miner Res. 2002. PMID: 12054173
-
11beta-Hydroxysteroid dehydrogenase expression and glucocorticoid synthesis are directed by a molecular switch during osteoblast differentiation.Mol Endocrinol. 2005 Mar;19(3):621-31. doi: 10.1210/me.2004-0212. Epub 2004 Dec 9. Mol Endocrinol. 2005. PMID: 15591536
-
11β-Hydroxysteroid dehydrogenase type 1: relevance of its modulation in the pathophysiology of obesity, the metabolic syndrome and type 2 diabetes mellitus.Diabetes Obes Metab. 2012 Oct;14(10):869-81. doi: 10.1111/j.1463-1326.2012.01582.x. Epub 2012 Mar 8. Diabetes Obes Metab. 2012. PMID: 22321826 Review.
-
11beta-hydroxysteroid dehydrogenase type 1: a tissue-specific regulator of glucocorticoid response.Endocr Rev. 2004 Oct;25(5):831-66. doi: 10.1210/er.2003-0031. Endocr Rev. 2004. PMID: 15466942 Review.
Cited by
-
Regulation of 11β-HSD1 reductase and the HPA axis by long-snake moxibustion in kidney-yang deficiency rats.Heliyon. 2024 Sep 30;10(19):e38486. doi: 10.1016/j.heliyon.2024.e38486. eCollection 2024 Oct 15. Heliyon. 2024. PMID: 39397979 Free PMC article.
References
-
- Andrzejewska A., Catar R., Schoon J., Qazi T. H., Sass F. A., Jacobi D., et al. (2019). Multi-parameter analysis of biobanked human bone marrow stromal cells shows little influence for donor age and mild comorbidities on phenotypic and functional properties. Front. Immunol. 10, 2474. 10.3389/fimmu.2019.02474 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

