Multi-omic analyses identify mucosa bacteria and fecal metabolites associated with weight loss after fecal microbiota transplantation
- PMID: 36091491
- PMCID: PMC9460156
- DOI: 10.1016/j.xinn.2022.100304
Multi-omic analyses identify mucosa bacteria and fecal metabolites associated with weight loss after fecal microbiota transplantation
Abstract
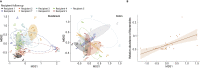
Fecal microbiota transplantation (FMT) has shown promising results in animal models of obesity, while results in human studies are inconsistent. We aimed to determine factors associated with weight loss after FMT in nine obese subjects using serial multi-omics analysis of the fecal and mucosal microbiome. The mucosal microbiome, fecal microbiome, and fecal metabolome showed individual clustering in each subject after FMT. The colonic microbiome in patients showed more marked variance after FMT compared with the duodenal microbiome, characterized by an increased relative abundance of Bacteroides. Subjects who lost weight after FMT sustained enrichment of Bifidobacterium bifidum and Alistipes onderdonkii in the duodenal, colonic mucosal, and fecal microbiome and increased levels of phosphopantothenate biosynthesis and fecal metabolite eicosapentaenoic acid (EPA), compared with those without weight loss. Fecal levels of amino acid metabolism-associated were positively correlated with the fecal abundance of B. bifidum, and fatty acid metabolism-associated metabolites showed positive correlations with A. onderdonkii. We report for the first time the individualized response of fecal and mucosa microbiome to FMT in obese subjects and highlight that FMT is less capable of shaping the small intestine microbiota. These findings contribute to personalized microbe-based therapies for obesity.
Keywords: Fecal microbiota transplantation; fecal metabolome; fecal microbiome; mucosal microbiome; obesity.
© 2022 The Author(s).
Conflict of interest statement
FKLC and SCN are the scientific co-founders and sit on the board of Directors of GenieBiome Ltd. SCN has served as an advisory board member for Pfizer, Ferring, Janssen, and Abbvie and a speaker for Ferring, Tillotts, Menarini, Janssen, Abbvie, and Takeda. She has received research grants from Olympus, Ferring, and Abbvie. FKLC has served as an advisor and lecture speaker for Eisai Co. Ltd., AstraZeneca, Pfizer Inc., Takeda Pharmaceutical Co., and Takeda (China) Holdings Co. Ltd. ZX and WT are part-time employee of GenieBiome Ltd. SCN, FKLC, TZ and ZX are inventors on patent held by the CUHK and MagIC that covers use of microorganisms in bodyweight regulation. All other co-authors have no conflict of interest.
Figures





References
-
- Lee P., Yacyshyn B.R., Yacyshyn M.B. Gut microbiota and obesity: an opportunity to alter obesity through faecal microbiota transplant (FMT) Diabetes Obes. Metab. 2019;21:479–490. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

