Anti-Inflammatory Effects and Mechanisms of Dandelion in RAW264.7 Macrophages and Zebrafish Larvae
- PMID: 36091818
- PMCID: PMC9454954
- DOI: 10.3389/fphar.2022.906927
Anti-Inflammatory Effects and Mechanisms of Dandelion in RAW264.7 Macrophages and Zebrafish Larvae
Abstract
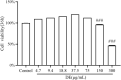
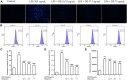
Dandelions (Taraxacum spp.) play an important role in the treatment of inflammatory diseases. In this study, we investigated the anti-inflammatory effects of Dandelion Extract (DE) in LPS-induced RAW264.7 macrophages and copper sulfate (CuSO4)-induced zebrafish larvae. DE was not toxic to RAW264.7 cells at 75 μg/ml as measured by cell viability, and DE inhibited LPS-induced cell morphological changes as measured by inverted microscopy. In survival experiments, DE at 25 μg/ml had no toxicity to zebrafish larvae. By using an enzymatic standard assay, DE reduced the production of nitric oxide (NO) in LPS-induced RAW264.7 cells. Fluorescence microscopy results show that DE reduced LPS-induced ROS production and apoptosis in RAW264.7 cells. DE also inhibited CuSO4-induced ROS production and neutrophil aggregation in zebrafish larvae. The results of flow cytometry show that DE alleviated the LPS-induced cell cycle arrest. In LPS-induced RAW264.7 cells, RT-PCR revealed that DE decreased the expression of M1 phenotypic genes iNOS, IL-6, and IL-1β while increasing the expression of M2 phenotypic genes IL-10 and CD206. Furthermore, in CuSO4-induced zebrafish larvae, DE reduced the expression of iNOS, TNF-α, IL-6, and IL-10. The findings suggest that DE reduces the LPS-induced inflammatory response in RAW264.7 cells by regulating polarization and apoptosis. DE also reduces the CuSO4-induced inflammatory response in zebrafish larvae.
Keywords: M1/M2 subtype; RAW264.7 cells; apoptosis; dandelion extract (DE); inflammation; zebrafish larvae.
Copyright © 2022 Li, Luo, Wu, Fan, Yang, Zhong, Guan, Wang and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

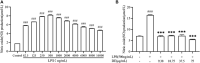





Similar articles
-
Taraxacum mongolicum extract inhibited malignant phenotype of triple-negative breast cancer cells in tumor-associated macrophages microenvironment through suppressing IL-10 / STAT3 / PD-L1 signaling pathways.J Ethnopharmacol. 2021 Jun 28;274:113978. doi: 10.1016/j.jep.2021.113978. Epub 2021 Mar 11. J Ethnopharmacol. 2021. PMID: 33716082
-
Anti-inflammatory effect and mechanism of stytontriterpene D on RAW264.7 cells and zebrafish.Front Pharmacol. 2025 Apr 28;16:1559022. doi: 10.3389/fphar.2025.1559022. eCollection 2025. Front Pharmacol. 2025. PMID: 40356999 Free PMC article.
-
Anti-inflammatory effects of 6S-5-methyltetrahydrofolate-calcium on RAW264.7 cells and zebrafish.Life Sci. 2023 Aug 15;327:121839. doi: 10.1016/j.lfs.2023.121839. Epub 2023 Jun 7. Life Sci. 2023. PMID: 37290666
-
Abrusamide H Impairs the Secretion of the Cytokines in RAW264.7 Cells and the Inflammatory Infiltration in Tail Transection-Induced Zebrafish.Chem Biodivers. 2022 Nov;19(11):e202200474. doi: 10.1002/cbdv.202200474. Epub 2022 Oct 17. Chem Biodivers. 2022. PMID: 36190475
-
Enoxaparin sodium bone cement plays an anti-inflammatory immunomodulatory role by inducing the polarization of M2 macrophages.J Orthop Surg Res. 2023 May 23;18(1):380. doi: 10.1186/s13018-023-03865-8. J Orthop Surg Res. 2023. PMID: 37221568 Free PMC article.
Cited by
-
Corydalis decumbens Alleviates the Migration, Phagocytosis, and Inflammatory Response of Macrophages.Evid Based Complement Alternat Med. 2023 Feb 22;2023:7000477. doi: 10.1155/2023/7000477. eCollection 2023. Evid Based Complement Alternat Med. 2023. PMID: 36874618 Free PMC article.
-
Phenethylferulate as a natural inhibitor of inflammation in LPS-stimulated RAW 264.7 macrophages: focus on NF-κB, Akt and MAPK signaling pathways.BMC Complement Med Ther. 2023 Nov 7;23(1):398. doi: 10.1186/s12906-023-04234-y. BMC Complement Med Ther. 2023. PMID: 37936108 Free PMC article.
-
Qinhuo Shanggan oral solution resolves acute lung injury by down-regulating TLR4/NF-κB signaling cascade and inhibiting NLRP3 inflammasome activation.Front Immunol. 2023 Oct 25;14:1285550. doi: 10.3389/fimmu.2023.1285550. eCollection 2023. Front Immunol. 2023. PMID: 37954597 Free PMC article.
-
The Galloyl Group Enhances the Inhibitory Activity of Catechins against LPS-Triggered Inflammation in RAW264.7 Cells.Foods. 2024 Aug 21;13(16):2616. doi: 10.3390/foods13162616. Foods. 2024. PMID: 39200543 Free PMC article.
-
Influence of Sulfur Fumigation on Angelicae Dahuricae Radix: Insights from Chemical Profiles, MALDI-MSI and Anti-Inflammatory Activities.Molecules. 2024 Dec 25;30(1):22. doi: 10.3390/molecules30010022. Molecules. 2024. PMID: 39795081 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

