Frontostriatal circuitry as a target for fMRI-based neurofeedback interventions: A systematic review
- PMID: 36092647
- PMCID: PMC9449529
- DOI: 10.3389/fnhum.2022.933718
Frontostriatal circuitry as a target for fMRI-based neurofeedback interventions: A systematic review
Abstract
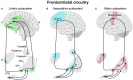
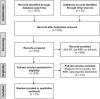
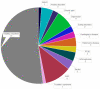
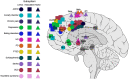
Dysregulated frontostriatal circuitries are viewed as a common target for the treatment of aberrant behaviors in various psychiatric and neurological disorders. Accordingly, experimental neurofeedback paradigms have been applied to modify the frontostriatal circuitry. The human frontostriatal circuitry is topographically and functionally organized into the "limbic," the "associative," and the "motor" subsystems underlying a variety of affective, cognitive, and motor functions. We conducted a systematic review of the literature regarding functional magnetic resonance imaging-based neurofeedback studies that targeted brain activations within the frontostriatal circuitry. Seventy-nine published studies were included in our survey. We assessed the efficacy of these studies in terms of imaging findings of neurofeedback intervention as well as behavioral and clinical outcomes. Furthermore, we evaluated whether the neurofeedback targets of the studies could be assigned to the identifiable frontostriatal subsystems. The majority of studies that targeted frontostriatal circuitry functions focused on the anterior cingulate cortex, the dorsolateral prefrontal cortex, and the supplementary motor area. Only a few studies (n = 14) targeted the connectivity of the frontostriatal regions. However, post-hoc analyses of connectivity changes were reported in more cases (n = 32). Neurofeedback has been frequently used to modify brain activations within the frontostriatal circuitry. Given the regulatory mechanisms within the closed loop of the frontostriatal circuitry, the connectivity-based neurofeedback paradigms should be primarily considered for modifications of this system. The anatomical and functional organization of the frontostriatal system needs to be considered in decisions pertaining to the neurofeedback targets.
Keywords: connectivity neurofeedback; frontostriatal circuitry; neurofeedback; neuromodulation; real-time fMRI.
Copyright © 2022 Orth, Meeh, Gur, Neuner and Sarkheil.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources

