Discovery of new symmetrical and asymmetrical nitrile-containing 1,4-dihydropyridine derivatives as dual kinases and P-glycoprotein inhibitors: synthesis, in vitro assays, and in silico studies
- PMID: 36093880
- PMCID: PMC9481151
- DOI: 10.1080/14756366.2022.2120478
Discovery of new symmetrical and asymmetrical nitrile-containing 1,4-dihydropyridine derivatives as dual kinases and P-glycoprotein inhibitors: synthesis, in vitro assays, and in silico studies
Abstract
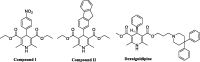
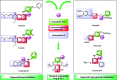
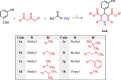
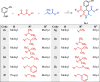
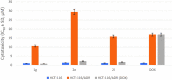
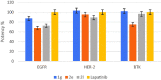
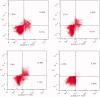
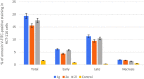
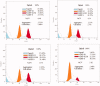
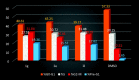
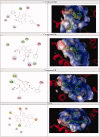
Two new series of symmetric (1a-h) and asymmetric (2a-l) 1,4-DHP derivatives were designed, synthesised, and evaluated as anticancer agents. In vitro anticancer screening of target compounds via National cancer institute "NCI" revealed that analogues 1g, 2e, and 2l demonstrated antiproliferative action with mean growth inhibition percentage "GI%" = 41, 28, and 64, respectively. The reversal doxorubicin (DOX) effects of compounds 1g, 2e, and 2l were examined and illustrated better cytotoxic activity with IC50 =1.12, 3.64, and 3.57 µM, respectively. The most active anticancer analogues, 1g, 2e, and 2l, were inspected for their putative mechanism of action by estimating their epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER-2), and Bruton's tyrosine kinase (BTK) inhibitory activities. Furthermore, the antimicrobial activity of target compounds was assessed against six different pathogens, followed by determining the minimum inhibitory concentration "MIC" values for the most active analogues. Molecular docking study was achieved to understand mode of interactions between selected inhibitors and different biological targets.
Keywords: 1,4-DHPs; P-glycoprotein; anticancer; antimicrobial; apoptosis; multidrug resistance (MDR); receptor tyrosine kinases (RTKs).
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures




References
-
- Chen S, Zhao Y, Liu S, et al. Epigenetic enzyme mutations as mediators of anti-cancer drug resistance. Drug Resistance Updates 2022;61:100821–13. - PubMed
-
- Li X, Li M, Huang M, et al. The multi-molecular mechanisms of tumor-targeted drug resistance in precision medicine. Biomed Pharmacother 2022;150:113064–15. - PubMed
-
- Friedlaender A, Subbiah V, Russo A, et al. EGFR and HER2 exon 20 insertions in solid tumours: from biology to treatment. Nat Rev Clin Oncol 2022;19:51–69. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
