Pf SRPK1 Regulates Asexual Blood Stage Schizogony and Is Essential for Male Gamete Formation
- PMID: 36094218
- PMCID: PMC9602455
- DOI: 10.1128/spectrum.02141-22
Pf SRPK1 Regulates Asexual Blood Stage Schizogony and Is Essential for Male Gamete Formation
Abstract
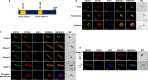
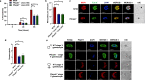
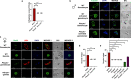
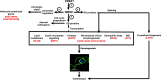
Serine/arginine-rich protein kinases (SRPKs) are cell cycle-regulated serine/threonine protein kinases and are important regulators of splicing factors. In this study, we functionally characterize SRPK1 of the human malaria parasite Plasmodium falciparum. P. falciparum SRPK1 (PfSRPK1) was expressed in asexual blood-stage and sexual-stage gametocytes. Pfsrpk1- parasites formed asexual schizonts that generated far fewer merozoites than wild-type parasites, causing reduced replication rates. Pfsrpk1- parasites also showed a severe defect in the differentiation of male gametes, causing a complete block in parasite transmission to mosquitoes. RNA sequencing (RNA-seq) analysis of wild-type PfNF54 and Pfsrpk1- stage V gametocytes suggested a role for PfSRPK1 in regulating transcript splicing and transcript abundance of genes coding for (i) microtubule/cilium morphogenesis-related proteins, (ii) proteins involved in cyclic nucleotide metabolic processes, (iii) proteins involved in signaling such as PfMAP2, (iv) lipid metabolism enzymes, (v) proteins of osmophilic bodies, and (vi) crystalloid components. Our study reveals an essential role for PfSRPK1 in parasite cell morphogenesis and suggests this kinase as a target to prevent malaria transmission from humans to mosquitoes. IMPORTANCE Plasmodium sexual stages represent a critical bottleneck in the parasite life cycle. Gametocytes taken up in an infectious blood meal by female anopheline mosquito get activated to form gametes and fuse to form short-lived zygotes, which transform into ookinetes to infect mosquitoes. In the present study, we demonstrate that PfSRPK1 is important for merozoite formation and critical for male gametogenesis and is involved in transcript homeostasis for numerous parasite genes. Targeting PfSRPK1 and its downstream pathways may reduce parasite replication and help achieve effective malaria transmission-blocking strategies.
Keywords: RNA-seq; SRPK1; exflagellation; gametocyte; mosquito; transmission.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

