Wearable Seismocardiography-Based Assessment of Stroke Volume in Congenital Heart Disease
- PMID: 36102243
- PMCID: PMC9683676
- DOI: 10.1161/JAHA.122.026067
Wearable Seismocardiography-Based Assessment of Stroke Volume in Congenital Heart Disease
Abstract
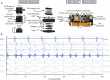
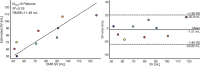
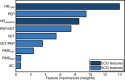
Background Patients with congenital heart disease (CHD) are at risk for the development of low cardiac output and other physiologic derangements, which could be detected early through continuous stroke volume (SV) measurement. Unfortunately, existing SV measurement methods are limited in the clinic because of their invasiveness (eg, thermodilution), location (eg, cardiac magnetic resonance imaging), or unreliability (eg, bioimpedance). Multimodal wearable sensing, leveraging the seismocardiogram, a sternal vibration signal associated with cardiomechanical activity, offers a means to monitoring SV conveniently, affordably, and continuously. However, it has not been evaluated in a population with significant anatomical and physiological differences (ie, children with CHD) or compared against a true gold standard (ie, cardiac magnetic resonance). Here, we present the feasibility of wearable estimation of SV in a diverse CHD population (N=45 patients). Methods and Results We used our chest-worn wearable biosensor to measure baseline ECG and seismocardiogram signals from patients with CHD before and after their routine cardiovascular magnetic resonance imaging, and derived features from the measured signals, predominantly systolic time intervals, to estimate SV using ridge regression. Wearable signal features achieved acceptable SV estimation (28% error with respect to cardiovascular magnetic resonance imaging) in a held-out test set, per cardiac output measurement guidelines, with a root-mean-square error of 11.48 mL and R2 of 0.76. Additionally, we observed that using a combination of electrical and cardiomechanical features surpassed the performance of either modality alone. Conclusions A convenient wearable biosensor that estimates SV enables remote monitoring of cardiac function and may potentially help identify decompensation in patients with CHD.
Keywords: cardiac output; machine learning; multimodal; noninvasive; pediatrics.
Figures

References
-
- Klem I, Shah DJ, White RD, Pennell DJ, van Rossum AC, Regenfus M, Sechtem U, Schvartzman PR, Hunold P, Croisille P, et al. Prognostic value of routine cardiac magnetic resonance assessment of left ventricular ejection fraction and myocardial damage. Circ Cardiovasc Imaging. 2011;4:610–619. doi: 10.1161/CIRCIMAGING.111.964965 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

