Maternal immune activation induces autism-like changes in behavior, neuroinflammatory profile and gut microbiota in mouse offspring of both sexes
- PMID: 36104346
- PMCID: PMC9474453
- DOI: 10.1038/s41398-022-02149-9
Maternal immune activation induces autism-like changes in behavior, neuroinflammatory profile and gut microbiota in mouse offspring of both sexes
Abstract
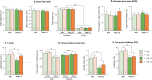
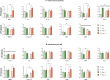
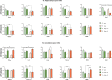
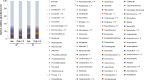
Autism Spectrum Disorder (ASD) is a sex-biased neurodevelopmental disorder with a male to female prevalence of 4:1, characterized by persistent deficits in social communication and interaction and restricted-repetitive patterns of behavior, interests or activities. Microbiota alterations as well as signs of neuroinflammation have been also reported in ASD. The involvement of immune dysregulation in ASD is further supported by evidence suggesting that maternal immune activation (MIA), especially during early pregnancy, may be a risk factor for ASD. The present study was aimed at characterizing the effects of MIA on behavior, gut microbiota and neuroinflammation in the mouse offspring also considering the impact of MIA in the two sexes. MIA offspring exhibited significant ASD-like behavioral alterations (i.e., deficits in sociability and sensorimotor gating, perseverative behaviors). The analysis of microbiota revealed changes in specific microbial taxa that recapitulated those seen in ASD children. In addition, molecular analyses indicated sex-related differences in the neuroinflammatory responses triggered by MIA, with a more prominent effect in the cerebellum. Our data suggest that both sexes should be included in the experimental designs of preclinical studies in order to identify those mechanisms that confer different vulnerability to ASD to males and females.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Association AP. Diagnostic and statistical manual of mental disorders. 5th ed. USA: CBS; 2013.

