Preparation of Fe3O4@SW-MIL-101-NH2 for selective pre-concentration of chlorogenic acid metabolites in rat plasma, urine, and feces samples
- PMID: 36105170
- PMCID: PMC9463528
- DOI: 10.1016/j.jpha.2022.01.002
Preparation of Fe3O4@SW-MIL-101-NH2 for selective pre-concentration of chlorogenic acid metabolites in rat plasma, urine, and feces samples
Abstract
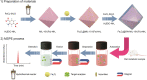
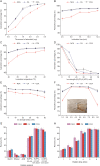
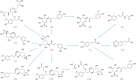
An innovative sandwich-structural Fe-based metal-organic framework magnetic material (Fe3O4@SW-MIL-101-NH2) was fabricated using a facile solvothermal method. The characteristic properties of the material were investigated by field emission scanning electron microscopy, transmission electron microscopy (TEM), energy-dispersive X-ray spectroscopy, Fourier transform infrared spectroscopy, X-ray powder diffraction, vibrating sample magnetometry, and Brunauer-Emmett-Teller measurements. Fe3O4@SW-MIL-101-NH2 is associated with advantages, such as robust magnetic properties, high specific surface area, and satisfactory storage stability, as well as good selective recognition ability for chlorogenic acid (CA) and its metabolites via chelation, hydrogen bonding, and π-interaction. The results of the static adsorption experiment indicated that Fe3O4@SW-MIL-101-NH2 possessed a high adsorption capacity toward CA and its isomers, cryptochlorogenic acid (CCA) and neochlorogenic acid (NCA), and the adsorption behaviors were fitted using the Langmuir adsorption isotherm model. Then, a strategy using magnetic solid-phase extraction (MSPE) and ultra-performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry (UPLC-Q-TOF MS/MS) was developed and successfully employed for the selective pre-concentration and rapid identification of CA metabolites in rat plasma, urine, and feces samples. This work presents a prospective strategy for the synthesis of magnetic adsorbents and the high-efficiency pretreatment of CA metabolites.
Keywords: Chlorogenic acid; Magnetic solid-phase extraction; Metabolic pathway; Metal-organic framework; Sandwich structure.
© 2022 The Authors.
Figures


References
-
- Nakatani N., Kayano S., Kikuzaki H., et al. Identification, quantitative determination, and antioxidative activities of chlorogenic acid isomers in prune (Prunus domestica L.) J. Agric. Food Chem. 2000;48:5512–5516. - PubMed
-
- Li N., Gao X.-Y., Fan Q., et al. Rapid determination of chlorogenic acid in aqueous solution of Flos Lonicerae Japonicae extraction. Zhongguo Zhongyao Zazhi. 2007;32:312–314. - PubMed
-
- Arai K., Terashima H., Aizawa S., et al. Simultaneous determination of trigonelline, caffeine, chlorogenic acid and their related compounds in instant coffee samples by HPLC using an acidic mobile phase containing octanesulfonate. Anal. Sci. 2015;31:831–835. - PubMed
-
- Sato Y., Itagaki S., Kurokawa T., et al. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharm. 2011;403:136–138. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

