Clinical Use of Tecovirimat (Tpoxx) for Treatment of Monkeypox Under an Investigational New Drug Protocol - United States, May-August 2022
- PMID: 36107794
- PMCID: PMC9484807
- DOI: 10.15585/mmwr.mm7137e1
Clinical Use of Tecovirimat (Tpoxx) for Treatment of Monkeypox Under an Investigational New Drug Protocol - United States, May-August 2022
Abstract
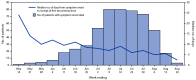
Currently, no Food and Drug Administration (FDA)-approved treatments for human monkeypox are available. Tecovirimat (Tpoxx), however, is an antiviral drug that has demonstrated efficacy in animal studies and is FDA-approved for treating smallpox. Use of tecovirimat for treatment of monkeypox in the United States is permitted only through an FDA-regulated Expanded Access Investigational New Drug (EA-IND) mechanism. CDC holds a nonresearch EA-IND protocol that facilitates access to and use of tecovirimat for treatment of monkeypox.§ The protocol includes patient treatment and adverse event reporting forms to monitor safety and ensure intended clinical use in accordance with FDA EA-IND requirements. The current multinational monkeypox outbreak, first detected in a country where Monkeypox virus infection is not endemic in May 2022, has predominantly affected gay, bisexual, and other men who have sex with men (MSM) (1,2). To describe characteristics of persons treated with tecovirimat for Monkeypox virus infection, demographic and clinical data abstracted from available tecovirimat EA-IND treatment forms were analyzed. As of August 20, 2022, intake and outcome forms were available for 549 and 369 patients, respectively; 97.7% of patients were men, with a median age of 36.5 years. Among patients with available data, 38.8% were reported to be non-Hispanic White (White) persons, 99.8% were prescribed oral tecovirimat, and 93.1% were not hospitalized. Approximately one half of patients with Monkeypox virus infection who received tecovirimat were living with HIV infection. The median interval from initiation of tecovirimat to subjective improvement was 3 days and did not differ by HIV infection status. Adverse events were reported in 3.5% of patients; all but one adverse event were nonserious. These data support the continued access to and treatment with tecovirimat for patients with or at risk for severe disease in the ongoing monkeypox outbreak.
Conflict of interest statement
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. Riad Elmor reports contract support (DCIPHER Program Management and Data Analytic Services) from Booz Allen Hamilton. No other potential conflicts of interest were disclosed.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

