Three-dimensional functional anatomy of the human sinoatrial node for epicardial and endocardial mapping and ablation
- PMID: 36113768
- PMCID: PMC9897959
- DOI: 10.1016/j.hrthm.2022.08.039
Three-dimensional functional anatomy of the human sinoatrial node for epicardial and endocardial mapping and ablation
Abstract
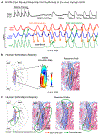
The sinoatrial node (SAN) is the primary pacemaker of the human heart. It is a single, elongated, 3-dimensional (3D) intramural fibrotic structure located at the junction of the superior vena cava intercaval region bordering the crista terminalis (CT). SAN activation originates in the intranodal pacemakers and is conducted to the atria through 1 or more discrete sinoatrial conduction pathways. The complexity of the 3D SAN pacemaker structure and intramural conduction are underappreciated during clinical multielectrode mapping and ablation procedures of SAN and atrial arrhythmias. In fact, defining and targeting SAN is extremely challenging because, even during sinus rhythm, surface-only multielectrode mapping may not define the leading pacemaker sites in intramural SAN but instead misinterpret them as epicardial or endocardial exit sites through sinoatrial conduction pathways. These SAN exit sites may be distributed up to 50 mm along the CT beyond the ∼20-mm-long anatomic SAN structure. Moreover, because SAN reentrant tachycardia beats may exit through the same sinoatrial conduction pathway as during sinus rhythm, many SAN arrhythmias are underdiagnosed. Misinterpretation of arrhythmia sources and/or mechanisms (eg, enhanced automaticity, intranodal vs CT reentry) limits diagnosis and success of catheter ablation treatments for poorly understood SAN arrhythmias. The aim of this review is to provide a state-of-the-art overview of the 3D structure and function of the human SAN complex, mechanisms of SAN arrhythmias and available approaches for electrophysiological mapping, 3D structural imaging, pharmacologic interventions, and ablation to improve diagnosis and mechanistic treatment of SAN and atrial arrhythmias.
Keywords: Ablation; Adenosine; Crista terminalis; Optical mapping; Reentry; Sinoatrial node; Sinus tachycardia; Three-dimensional electroanatomic mapping.
Copyright © 2022 Heart Rhythm Society. Published by Elsevier Inc. All rights reserved.
Figures






References
-
- Shimamoto K, Yamagata K, Nakajima K, et al. An anatomical approach to determine the location of the sinoatrial node during catheter ablation. J Cardiovasc Electrophysiol. 2021;32:1320–1327. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

