Market survey of disposable e-cigarette nicotine content and e-liquid volume
- PMID: 36114568
- PMCID: PMC9482302
- DOI: 10.1186/s12889-022-14152-2
Market survey of disposable e-cigarette nicotine content and e-liquid volume
Abstract
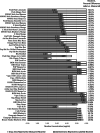
Inaccurate labels on some e-cigarette products have prompted calls for routine testing to monitor product label integrity. The objective of this study was to compare label statements of commercial disposable/non-chargeable e-cigarette products for nicotine concentration and e-liquid volume with analytically verified levels. Commercial e-cigarette samples were analyzed for nicotine concentration (N = 51), e-liquid volume and total nicotine content (N = 39). Twenty-three of the 51 samples analyzed for nicotine deviated from their label statements by more than ± 10%. Deviations ranged from -50.1% to + 13.9%. Thirty of the 39 samples analyzed for e-liquid volume deviated from their label statements by more than ± 10%. Deviations ranged from -62.1% to + 13.3%. Only one brand listed total nicotine on the label. In thirty-one of the 39 samples, calculated total nicotine amount in e-liquid deviated from the amounts calculated from the label metrics by more than ± 10%. Deviations ranged from -66.8% to -1.43%. These findings underscore the need for regulatory enforcement of manufacturing quality control and product labeling practices to optimize the harm reduction potential and consumer experience associated with the use of e-cigarette products.
Keywords: Nicotine levels; Product labeling; e-cigarettes.
© 2022. The Author(s).
Conflict of interest statement
Willie McKinney is an independent consultant contracted by tobacco product manufacturers to provide scientific and regulatory support. Scott Appleton, Karin Gilligan, Helen Cyrus-Miller, and Ryan Seltzer are independent consultants contracted by McKinney Regulatory Science Advisors, LLC to design and execute scientific studies, in addition to drafting and reviewing manuscripts. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- National Academies of Sciences, E.a.M . Public Health Consequences of E-Cigarettes. Washington: The National Academies Press; 2018. p. 774. - PubMed
-
- Foulds J, Cobb CO, Yen M-S, Veldheer S, Brosnan P, Yingst J, Hrabovsky S, Lopez AA, Allen SI, Bullen C, et al. Effect of Electronic Nicotine Delivery Systems on Cigarette Abstinence in Smokers with no Plans to Quit: Exploratory Analysis of a Randomized Placebo-Controlled Trial. 2021 10.1101/2021.06.22.21259359. - PMC - PubMed
-
- U.S. Food and Drug Adminstration [FDA]. FDA: Foods Must Contain What Label Says. Available online: https://www.fda.gov/consumers/consumer-updates/fda-foods-must-contain-wh... (Accessed 20 Nov 2021).
-
- Congress, U.S. Family Smoking Prevention and Tobacco Control Act. 2009.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

