Plasma-induced nanoparticle aggregation for stratifying COVID-19 patients according to disease severity
- PMID: 36124254
- PMCID: PMC9476366
- DOI: 10.1016/j.snb.2022.132638
Plasma-induced nanoparticle aggregation for stratifying COVID-19 patients according to disease severity
Abstract
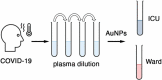
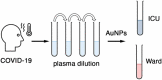
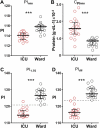
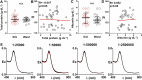
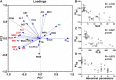
Stratifying patients according to disease severity has been a major hurdle during the COVID-19 pandemic. This usually requires evaluating the levels of several biomarkers, which may be cumbersome when rapid decisions are required. In this manuscript we show that a single nanoparticle aggregation test can be used to distinguish patients that require intensive care from those that have already been discharged from the intensive care unit (ICU). It consists of diluting a platelet-free plasma sample and then adding gold nanoparticles. The nanoparticles aggregate to a larger extent when the samples are obtained from a patient in the ICU. This changes the color of the colloidal suspension, which can be evaluated by measuring the pixel intensity of a photograph. Although the exact factor or combination of factors behind the different aggregation behavior is unknown, control experiments demonstrate that the presence of proteins in the samples is crucial for the test to work. Principal component analysis demonstrates that the test result is highly correlated to biomarkers of prognosis and inflammation that are commonly used to evaluate the severity of COVID-19 patients. The results shown here pave the way to develop nanoparticle aggregation assays that classify COVID-19 patients according to disease severity, which could be useful to de-escalate care safely and make a better use of hospital resources.
Keywords: AST, aspartate aminotransferaseALT, alanine aminotransferase; Alb, albumin; C1.75, protein concentration 1.75 × 10-4 g·dL-1; CPImin, protein concentration at PImin; CRP, C-reactive protein; Colorimetric; Creat, creatinine; D-D, D-dimer; Ferr, ferritin; GGT, gamma-glutamyl transferase; Glu, glucose; Gold; Hb, hemoglobin; ICU, intensive care unit; INR, international normalized ratio (prothrombin time); LDH, lactate dehydrogenase; LSPR, localized surface plasmon resonance; MCV, mean corpuscular volume; MPV, mean platelet volume; Mono, monocytes; NIR, near-infrared; NLR, neutrophil-to-lymphocyte ratio; NTA, nanoparticle tracking analysis; PDW, platelet distribution width; PI, pixel intensity; PI1.75, pixel intensity at C1.75; PIdil, pixel intensity at plasma dilution 1:31250; PImin, minimum value of pixel intensity; PLR, platelet-to-lymphocyte ratio; Plasmonic; RBC, red blood cells; RDW, red cell distribution width; SARS-CoV-2; Sepsis; TG, triglycerides; TotProt, total protein concentration; WBC, white blood cells.
© 2022 Elsevier B.V. All rights reserved.
Conflict of interest statement
There are no conflicts of interest.
Figures
Similar articles
-
Lymphocyte-C-reactive protein ratio can differentiate disease severity of COVID-19 patients and serve as an assistant screening tool for hospital and ICU admission.Front Immunol. 2022 Sep 23;13:957407. doi: 10.3389/fimmu.2022.957407. eCollection 2022. Front Immunol. 2022. PMID: 36248811 Free PMC article.
-
SARS-CoV-2 Infection-Dependent Modulation in Vital Components of the Serum Profile of Severely SARS-CoV-2 Infected Patients.Infect Drug Resist. 2024 Apr 29;17:1653-1667. doi: 10.2147/IDR.S463238. eCollection 2024. Infect Drug Resist. 2024. PMID: 38707987 Free PMC article.
-
How do routine laboratory tests change in coronavirus disease 2019?Scand J Clin Lab Invest. 2021 Feb;81(1):24-33. doi: 10.1080/00365513.2020.1855470. Epub 2020 Dec 20. Scand J Clin Lab Invest. 2021. PMID: 33342313
-
A Review of Routine Laboratory Biomarkers for the Detection of Severe COVID-19 Disease.Int J Anal Chem. 2022 Oct 11;2022:9006487. doi: 10.1155/2022/9006487. eCollection 2022. Int J Anal Chem. 2022. PMID: 36267156 Free PMC article. Review.
-
Laboratory features of severe vs. non-severe COVID-19 patients in Asian populations: a systematic review and meta-analysis.Eur J Med Res. 2020 Aug 3;25(1):30. doi: 10.1186/s40001-020-00432-3. Eur J Med Res. 2020. PMID: 32746929 Free PMC article.
Cited by
-
Coronavirus Anatomy and Its Analytical Approaches for Targeting COVID-19.Adv Exp Med Biol. 2024;1457:33-44. doi: 10.1007/978-3-031-61939-7_2. Adv Exp Med Biol. 2024. PMID: 39283419 Review.
-
Effect of nanoshell geometries, sizes, and quantum emitter parameters on the sensitivity of plasmon-exciton hybrid nanoshells for sensing application.Sci Rep. 2023 Jul 13;13(1):11325. doi: 10.1038/s41598-023-38475-1. Sci Rep. 2023. PMID: 37443203 Free PMC article.
References
-
- Vesga M.J., McKechnie D., Laing S., Kearns H., Faulds K., Johnston K., Sefcik J. Effect of glycine on aggregation of citrate-functionalised gold nanoparticles and SERS measurements. Colloids Surf. A Physicochem. Eng. Asp. 2021;621 doi: 10.1016/j.colsurfa.2021.126523. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

