New antiretroviral inhibitors and HIV-1 drug resistance: more focus on 90% HIV-1 isolates?
- PMID: 36130204
- PMCID: PMC9841967
- DOI: 10.1093/femsre/fuac040
New antiretroviral inhibitors and HIV-1 drug resistance: more focus on 90% HIV-1 isolates?
Abstract
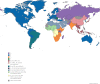
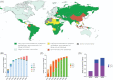
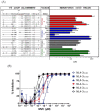
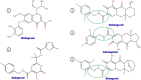
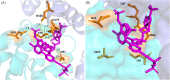
Combined HIV antiretroviral therapy (cART) has been effective except if drug resistance emerges. As cART has been rolled out in low-income countries, drug resistance has emerged at higher rates than observed in high income countries due to factors including initial use of these less tolerated cART regimens, intermittent disruptions in drug supply, and insufficient treatment monitoring. These socioeconomic factors impacting drug resistance are compounded by viral mechanistic differences by divergent HIV-1 non-B subtypes compared to HIV-1 subtype B that largely infects the high-income countries (just 10% of 37 million infected). This review compares the inhibition and resistance of diverse HIV-1 subtypes and strains to the various approved drugs as well as novel inhibitors in clinical trials. Initial sequence variations and differences in replicative fitness between HIV-1 subtypes pushes strains through different fitness landscapes to escape from drug selective pressure. The discussions here provide insight to patient care givers and policy makers on how best to use currently approved ART options and reduce the emergence of drug resistance in ∼33 million individuals infected with HIV-1 subtype A, C, D, G, and recombinants forms. Unfortunately, over 98% of the literature on cART resistance relates to HIV-1 subtype B.
Keywords: HIV-1 genetic diversity; HIV-1 subtype specific resistance; antiretroviral therapy; discovery of novel HIV inhibitors; low-and middle-income countries; non-subtype B HIV-1.
© The Author(s) 2022. Published by Oxford University Press on behalf of FEMS.
Figures




References
-
- Achieng L, Riedel DJ. Dolutegravir resistance and failure in a kenyan patient. J Infect Dis. 2019;219:165–7. - PubMed
-
- Ackerman P, Aberg J, Molina JMet al. . A Subgroup Analysis of the Week 96 Efficacy and Safety Results Evaluating Fostemsavir in Heavily Treatment-Experienced HIV-1 Infected Participants in the Phase 3 BRIGHTE Study: Results From The Randomized Cohort. IAS 2019: Conference on HIV Pathogenesis Treatment and Prevention. Mexico City, 2019. https://www.natap.org/2019/IAS/IAS_20.htm (10 April 2022, date last accessed).
-
- Aidsmap . Dolutegravir recommended for all in new World Health Organization guidelines. 2019. https://www.aidsmap.com/news/jul-2019/dolutegravir-recommended-all-new-w... (11 April 2022, date last accessed).
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

