The architecture of clonal expansions in morphologically normal tissue from cancerous and non-cancerous prostates
- PMID: 36131292
- PMCID: PMC9494848
- DOI: 10.1186/s12943-022-01644-3
The architecture of clonal expansions in morphologically normal tissue from cancerous and non-cancerous prostates
Abstract
Background: Up to 80% of cases of prostate cancer present with multifocal independent tumour lesions leading to the concept of a field effect present in the normal prostate predisposing to cancer development. In the present study we applied Whole Genome DNA Sequencing (WGS) to a group of morphologically normal tissue (n = 51), including benign prostatic hyperplasia (BPH) and non-BPH samples, from men with and men without prostate cancer. We assess whether the observed genetic changes in morphologically normal tissue are linked to the development of cancer in the prostate.
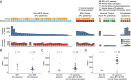
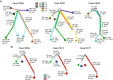
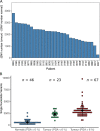
Results: Single nucleotide variants (P = 7.0 × 10-03, Wilcoxon rank sum test) and small insertions and deletions (indels, P = 8.7 × 10-06) were significantly higher in morphologically normal samples, including BPH, from men with prostate cancer compared to those without. The presence of subclonal expansions under selective pressure, supported by a high level of mutations, were significantly associated with samples from men with prostate cancer (P = 0.035, Fisher exact test). The clonal cell fraction of normal clones was always higher than the proportion of the prostate estimated as epithelial (P = 5.94 × 10-05, paired Wilcoxon signed rank test) which, along with analysis of primary fibroblasts prepared from BPH specimens, suggests a stromal origin. Constructed phylogenies revealed lineages associated with benign tissue that were completely distinct from adjacent tumour clones, but a common lineage between BPH and non-BPH morphologically normal tissues was often observed. Compared to tumours, normal samples have significantly less single nucleotide variants (P = 3.72 × 10-09, paired Wilcoxon signed rank test), have very few rearrangements and a complete lack of copy number alterations.
Conclusions: Cells within regions of morphologically normal tissue (both BPH and non-BPH) can expand under selective pressure by mechanisms that are distinct from those occurring in adjacent cancer, but that are allied to the presence of cancer. Expansions, which are probably stromal in origin, are characterised by lack of recurrent driver mutations, by almost complete absence of structural variants/copy number alterations, and mutational processes similar to malignant tissue. Our findings have implications for treatment (focal therapy) and early detection approaches.
Keywords: Benign prostatic hyperplasia; Clonal expansions; Field effect; Genomics; Mutational signatures; Normal tissue; Prostate cancer.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

