Rapid 13C Hyperpolarization of the TCA Cycle Intermediate α-Ketoglutarate via SABRE-SHEATH
- PMID: 36136056
- PMCID: PMC9907724
- DOI: 10.1021/acs.analchem.2c02160
Rapid 13C Hyperpolarization of the TCA Cycle Intermediate α-Ketoglutarate via SABRE-SHEATH
Abstract
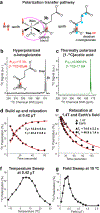
α-Ketoglutarate is a key biomolecule involved in a number of metabolic pathways─most notably the TCA cycle. Abnormal α-ketoglutarate metabolism has also been linked with cancer. Here, isotopic labeling was employed to synthesize [1-13C,5-12C,D4]α-ketoglutarate with the future goal of utilizing its [1-13C]-hyperpolarized state for real-time metabolic imaging of α-ketoglutarate analytes and its downstream metabolites in vivo. The signal amplification by reversible exchange in shield enables alignment transfer to heteronuclei (SABRE-SHEATH) hyperpolarization technique was used to create 9.7% [1-13C] polarization in 1 minute in this isotopologue. The efficient 13C hyperpolarization, which utilizes parahydrogen as the source of nuclear spin order, is also supported by favorable relaxation dynamics at 0.4 μT field (the optimal polarization transfer field): the exponential 13C polarization buildup constant Tb is 11.0 ± 0.4 s whereas the 13C polarization decay constant T1 is 18.5 ± 0.7 s. An even higher 13C polarization value of 17.3% was achieved using natural-abundance α-ketoglutarate disodium salt, with overall similar relaxation dynamics at 0.4 μT field, indicating that substrate deuteration leads only to a slight increase (∼1.2-fold) in the relaxation rates for 13C nuclei separated by three chemical bonds. Instead, the gain in polarization (natural abundance versus [1-13C]-labeled) is rationalized through the smaller heat capacity of the "spin bath" comprising available 13C spins that must be hyperpolarized by the same number of parahydrogen present in each sample, in line with previous 15N SABRE-SHEATH studies. Remarkably, the C-2 carbon was not hyperpolarized in both α-ketoglutarate isotopologues studied; this observation is in sharp contrast with previously reported SABRE-SHEATH pyruvate studies, indicating that the catalyst-binding dynamics of C-2 in α-ketoglutarate differ from that in pyruvate. We also demonstrate that 13C spectroscopic characterization of α-ketoglutarate and pyruvate analytes can be performed at natural 13C abundance with an estimated detection limit of 80 micromolar concentration × *%P13C. All in all, the fundamental studies reported here enable a wide range of research communities with a new hyperpolarized contrast agent potentially useful for metabolic imaging of brain function, cancer, and other metabolically challenging diseases.
Conflict of interest statement
Conflict of Interest Statement
BMG, EYC declare stake ownership in XeUS Technologies, LTD. TT holds stock in Vizma Life Sciences LLC.
Figures




References
-
- Hoult DI; Richards RE The signal-to-noise ratio of the nuclear magnetic resonance experiment. J. Magn. Reson. 1976, 24, 71–85. - PubMed
-
- Bluml S; Moreno-Torres A; Shic F; Nguy CH; Ross BD Tricarboxylic acid cycle of glia in the in vivo human brain. NMR Biomed. 2002, 15, 1–5. - PubMed
-
- Day SE; Kettunen MI; Gallagher FA; Hu DE; Lerche M; Wolber J; Golman K; Ardenkjaer-Larsen JH; Brindle KM Detecting tumor response to treatment using hyperpolarized C-13 magnetic resonance imaging and spectroscopy. Nat. Med. 2007, 13, 1382–1387. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

