Proinflammatory and Cancer-Promoting Pathobiont Fusobacterium nucleatum Directly Targets Colorectal Cancer Stem Cells
- PMID: 36139097
- PMCID: PMC9496236
- DOI: 10.3390/biom12091256
Proinflammatory and Cancer-Promoting Pathobiont Fusobacterium nucleatum Directly Targets Colorectal Cancer Stem Cells
Abstract
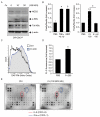
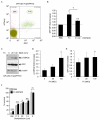
Intestinal bacterial communities participate in gut homeostasis and are recognized as crucial in bowel inflammation and colorectal cancer (CRC). Fusobacterium nucleatum (Fn), a pathobiont of the oral microflora, has recently emerged as a CRC-associated microbe linked to disease progression, metastasis, and a poor clinical outcome; however, the primary cellular and/or microenvironmental targets of this agent remain elusive. We report here that Fn directly targets putative colorectal cancer stem cells (CR-CSCs), a tumor cell subset endowed with cancer re-initiating capacity after surgery and chemotherapy. A patient-derived CSC line, highly enriched (70%) for the stem marker CD133, was expanded as tumor spheroids, dissociated, and exposed in vitro to varying amounts (range 100-500 MOI) of Fn. We found that Fn stably adheres to CSCs, likely by multiple interactions involving the tumor-associated Gal-GalNac disaccharide and the Fn-docking protein CEA-family cell adhesion molecule 1 (CEACAM-1), robustly expressed on CSCs. Importantly, Fn elicited innate immune responses in CSCs and triggered a growth factor-like, protein tyrosine phosphorylation cascade largely dependent on CEACAM-1 and culminating in the activation of p42/44 MAP kinase. Thus, the direct stimulation of CSCs by Fn may contribute to microbiota-driven colorectal carcinogenesis and represent a target for innovative therapies.
Keywords: PTPase; bacterial adhesins; cancer stem cells; carcino-embryonic antigen cell adhesion molecule-1; colorectal cancer; fusobacterium nucleatum; microbiota; tumor microenvironment; tumor spheroids.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Argilés G., Tabernero J., Labianca R., Hochhauser D., Salazar R., Iveson T., Laurent-Puig P., Quirke P., Yoshino T., Taieb J., et al. Localised Colon Cancer: ESMO Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2020;31:1291–1305. doi: 10.1016/j.annonc.2020.06.022. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

