Impact of Antioxidant Feed and Growth Manipulation on the Redox Regulation of Atlantic Salmon Smolts
- PMID: 36139780
- PMCID: PMC9495322
- DOI: 10.3390/antiox11091708
Impact of Antioxidant Feed and Growth Manipulation on the Redox Regulation of Atlantic Salmon Smolts
Abstract
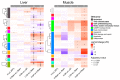
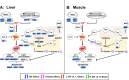
Accumulating evidence indicates a close relationship between oxidative stress and growth rate in fish. However, the underlying mechanisms of this relationship remain unclear. This study evaluated the combined effect of dietary antioxidants and growth hormone (GH) on the liver and the muscle redox status of Atlantic salmon. There were two sequential experimental phases (EP) termed EP1 and EP2, each lasting for 6 weeks. In EP1, Atlantic salmon were fed either low-(L, 230 mg/kg ascorbic acid (Asc), 120 mg/kg α-tocopherol (α-TOH)), or high-(H, 380 mg/kg Asc, 210 mg/kg α-TOH)vitamin diets. The vitamins were supplemented as stable forms and the feeding was continued in EP2. In EP2, half of the fish were implanted with 3 μL per g body weight of recombinant bovine GH (Posilac®, 1 mg rbGH g BW-1) suspended in sesame oil, while the other half were held in different tanks and sham-implanted with similar volumes of the sesame oil vehicle. Here, we show that increasing high levels of vitamin C and E (diet H) increased their content in muscle and liver during EP1. GH implantation decreased vitamin C and E levels in both liver and muscle but increased malondialdehyde (MDA) levels only in the liver. GH also affected many genes and pathways of antioxidant enzymes and the redox balance. Among the most consistent were the upregulation of genes coding for the NADPH oxidase family (NOXs) and downregulation of the oxidative stress response transcription factor, nuclear factor-erythroid 2-related factor 2 (nrf2), and its downstream target genes in the liver. We verified that GH increases the growth rate until the end of the trail and induces an oxidative effect in the liver and muscle of Atlantic salmon. Dietary antioxidants do lower oxidative stress but have no effect on the growth rate. The present study is intended as a starting point to understand the potential interactions between growth and redox signaling in fish.
Keywords: antioxidant nutrients; fish; growth hormone; oxidative stress; redox; transcriptional regulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


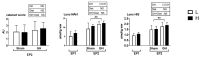




Similar articles
-
Seasonal Changes in Photoperiod: Effects on Growth and Redox Signaling Patterns in Atlantic Salmon Postsmolts.Antioxidants (Basel). 2023 Aug 2;12(8):1546. doi: 10.3390/antiox12081546. Antioxidants (Basel). 2023. PMID: 37627541 Free PMC article.
-
Antioxidant nutrition in Atlantic salmon (Salmo salar) parr and post-smolt, fed diets with high inclusion of plant ingredients and graded levels of micronutrients and selected amino acids.PeerJ. 2016 Nov 8;4:e2688. doi: 10.7717/peerj.2688. eCollection 2016. PeerJ. 2016. PMID: 27843721 Free PMC article.
-
Dietary alpha-tocopherol affects tissue vitamin e and malondialdehyde levels but does not change antioxidant enzymes and fatty acid composition in farmed Atlantic salmon (Salmo salar L.).Int J Vitam Nutr Res. 2013;83(4):238-45. doi: 10.1024/0300-9831/a000166. Int J Vitam Nutr Res. 2013. PMID: 25008014
-
Dietary resveratrol impairs body weight gain due to reduction of feed intake without affecting fatty acid composition in Atlantic salmon.Animal. 2019 Jan;13(1):25-32. doi: 10.1017/S1751731118000812. Epub 2018 Apr 23. Animal. 2019. PMID: 29681254
-
Effects of oxidised dietary fish oil and high-dose vitamin E supplementation on growth performance, feed utilisation and antioxidant defence enzyme activities of juvenile large yellow croaker (Larmichthys crocea).Br J Nutr. 2016 May;115(9):1531-8. doi: 10.1017/S0007114516000398. Epub 2016 Mar 7. Br J Nutr. 2016. PMID: 26948923
Cited by
-
Effect of Rearing, Physiological, and Processing Conditions on the Volatile Profile of Atlantic Salmon (Salmo salar) Using SIFT-MS.Foods. 2025 Jul 21;14(14):2540. doi: 10.3390/foods14142540. Foods. 2025. PMID: 40724360 Free PMC article.
-
Seasonal Changes in Photoperiod: Effects on Growth and Redox Signaling Patterns in Atlantic Salmon Postsmolts.Antioxidants (Basel). 2023 Aug 2;12(8):1546. doi: 10.3390/antiox12081546. Antioxidants (Basel). 2023. PMID: 37627541 Free PMC article.
References
-
- Alne H., Oehme M., Thomassen M., Terjesen B., Rorvik K.A. Reduced growth, condition factor and body energy levels in Atlantic salmon Salmo salar L. during their first spring in the sea. Aquac. Res. 2011;42:248–259. doi: 10.1111/j.1365-2109.2010.02618.x. - DOI
-
- Oppedal F., Berg A., Olsen R.E., Taranger G.L., Hansen T. Photoperiod in seawater influence seasonal growth and chemical composition in autumn sea-transferred Atlantic salmon (Salmo salar L.) given two vaccines. Aquaculture. 2006;254:396–410. doi: 10.1016/j.aquaculture.2005.10.026. - DOI
-
- Dessen J.E., Weihe R.N., Hatlen B., Thomassen M.S., Rorvik K.A. Different growth performance, lipid deposition, and nutrient utilization in in-season (S1) Atlantic salmon post-smolt fed isoenergetic diets differing in protein-to-lipid ratio. Aquaculture. 2017;473:345–354. doi: 10.1016/j.aquaculture.2017.02.006. - DOI
-
- Nordgarden U., Ørnsrud R., Hansen T., Hemre G.I. Seasonal changes in selected muscle quality parameters in Atlantic salmon (Salmo salar L.) reared under natural and continuous light. Aquac. Nutr. 2003;9:161–168. doi: 10.1046/j.1365-2095.2003.00236.x. - DOI
-
- Hamre K., Sissener N.H., Lock E.J., Olsvik P.A., Espe M., Torstensen B.E., Silva J., Johansen J., Waagbo R., Hemre G.I. Antioxidant nutrition in Atlantic salmon (Salmo salar) parr and post-smolt, fed diets with high inclusion of plant ingredients and graded levels of micronutrients and selected amino acids. PeerJ. 2016;4:e2688. doi: 10.7717/peerj.2688. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

