A Human Stem Cell-Derived Neurosensory-Epithelial Circuitry on a Chip to Model Herpes Simplex Virus Reactivation
- PMID: 36140168
- PMCID: PMC9495731
- DOI: 10.3390/biomedicines10092068
A Human Stem Cell-Derived Neurosensory-Epithelial Circuitry on a Chip to Model Herpes Simplex Virus Reactivation
Abstract
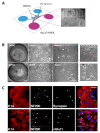
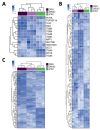
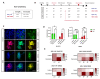
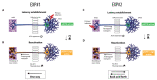
Both emerging viruses and well-known viral pathogens endowed with neurotropism can either directly impair neuronal functions or induce physio-pathological changes by diffusing from the periphery through neurosensory-epithelial connections. However, developing a reliable and reproducible in vitro system modeling the connectivity between the different human sensory neurons and peripheral tissues is still a challenge and precludes the deepest comprehension of viral latency and reactivation at the cellular and molecular levels. This study shows a stable topographic neurosensory-epithelial connection on a chip using human stem cell-derived dorsal root ganglia (DRG) organoids. Bulk and single-cell transcriptomics showed that different combinations of key receptors for herpes simplex virus 1 (HSV-1) are expressed by each sensory neuronal cell type. This neuronal-epithelial circuitry enabled a detailed analysis of HSV infectivity, faithfully modeling its dynamics and cell type specificity. The reconstitution of an organized connectivity between human sensory neurons and keratinocytes into microfluidic chips provides a powerful in vitro platform for modeling viral latency and reactivation of human viral pathogens.
Keywords: herpes simplex virus; keratinocytes; latency; microfluidics; organoids; reactivation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Herpes Simplex Virus 1 Strains 17syn+ and KOS(M) Differ Greatly in Their Ability To Reactivate from Human Neurons In Vitro.J Virol. 2020 Jul 16;94(15):e00796-20. doi: 10.1128/JVI.00796-20. Print 2020 Jul 16. J Virol. 2020. PMID: 32461310 Free PMC article.
-
An Immortalized Human Dorsal Root Ganglion Cell Line Provides a Novel Context To Study Herpes Simplex Virus 1 Latency and Reactivation.J Virol. 2017 May 26;91(12):e00080-17. doi: 10.1128/JVI.00080-17. Print 2017 Jun 15. J Virol. 2017. PMID: 28404842 Free PMC article.
-
Modulation of Voltage-Gated Sodium Channel Activity in Human Dorsal Root Ganglion Neurons by Herpesvirus Quiescent Infection.J Virol. 2020 Jan 17;94(3):e01823-19. doi: 10.1128/JVI.01823-19. Print 2020 Jan 17. J Virol. 2020. PMID: 31694955 Free PMC article.
-
A comparison of herpes simplex virus type 1 and varicella-zoster virus latency and reactivation.J Gen Virol. 2015 Jul;96(Pt 7):1581-602. doi: 10.1099/vir.0.000128. Epub 2015 Mar 20. J Gen Virol. 2015. PMID: 25794504 Free PMC article. Review.
-
Disturbed Yin-Yang balance: stress increases the susceptibility to primary and recurrent infections of herpes simplex virus type 1.Acta Pharm Sin B. 2020 Mar;10(3):383-398. doi: 10.1016/j.apsb.2019.06.005. Epub 2019 Jun 22. Acta Pharm Sin B. 2020. PMID: 32140387 Free PMC article. Review.
Cited by
-
Investigating epithelial-neuronal signaling contribution in visceral pain through colon organoid-dorsal root ganglion neuron co-cultures.Neural Regen Res. 2024 Jun 1;19(6):1199-1200. doi: 10.4103/1673-5374.386403. Epub 2023 Oct 2. Neural Regen Res. 2024. PMID: 37905863 Free PMC article. No abstract available.
-
Human brain microphysiological systems in the study of neuroinfectious disorders.Exp Neurol. 2023 Jul;365:114409. doi: 10.1016/j.expneurol.2023.114409. Epub 2023 Apr 14. Exp Neurol. 2023. PMID: 37061175 Free PMC article. Review.
-
iPSC-derived three-dimensional brain organoid models and neurotropic viral infections.J Neurovirol. 2023 Apr;29(2):121-134. doi: 10.1007/s13365-023-01133-3. Epub 2023 Apr 25. J Neurovirol. 2023. PMID: 37097597 Free PMC article. Review.
References
-
- BenMohamed L., Osorio N., Khan A.A., Srivastava R., Huang L., Krochmal J.J., Garcia J.M., Simpson J.L., Wechsler S.L. Prior Corneal Scarification and Injection of Immune Serum Are Not Required Before Ocular HSV-1 Infection for UV-B-Induced Virus Reactivation and Recurrent Herpetic Corneal Disease in Latently Infected Mice. Curr. Eye Res. 2016;41:747–756. doi: 10.3109/02713683.2015.1061024. - DOI - PMC - PubMed
-
- Chiuppesi F., Vannucci L., Luca A.D., Lai M., Matteoli B., Freer G., Manservigi R., Ceccherini-Nelli L., Maggi F., Bendinelli M., et al. A Lentiviral Vector-Based, Herpes Simplex Virus 1 (HSV-1) Glycoprotein B Vaccine Affords Cross-Protection against HSV-1 and HSV-2 Genital Infections. J. Virol. 2012;86:6563–6574. doi: 10.1128/JVI.00302-12. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

