β-Caryophyllene Counteracts Chemoresistance Induced by Cigarette Smoke in Triple-Negative Breast Cancer MDA-MB-468 Cells
- PMID: 36140359
- PMCID: PMC9496176
- DOI: 10.3390/biomedicines10092257
β-Caryophyllene Counteracts Chemoresistance Induced by Cigarette Smoke in Triple-Negative Breast Cancer MDA-MB-468 Cells
Abstract
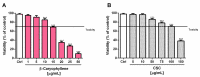
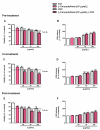
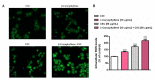
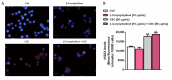
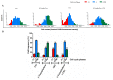
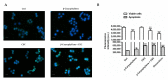
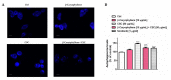
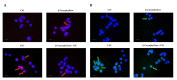
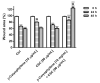
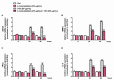
Exposure to cigarette smoke (CS) has been associated with an increased risk of fatal breast cancers and recurrence, along with chemoresistance and chemotherapy impairment. This strengthens the interest in chemopreventive agents to be exploited both in healthy and oncological subjects to prevent or repair CS damage. In the present study, we evaluated the chemopreventive properties of the natural sesquiterpene β-caryophyllene towards the damage induced by cigarette smoke condensate (CSC) in triple negative breast cancer MDA-MB-468 cells. Particularly, we assessed the ability of the sesquiterpene to interfere with the mechanisms exploited by CSC to promote cell survival and chemoresistance, including genomic instability, cell cycle progress, autophagy/apoptosis, cell migration and related pathways. β-Caryophyllene was found to be able to increase the CSC-induced death of MDA-MB-468 cells, likely triggering oxidative stress, cell cycle arrest and apoptosis; moreover, it hindered cell recovery, autophagy activation and cell migration; at last, a marked inhibition of the signal transducer and activator of transcription 3 (STAT3) activation was highlighted: this could represent a key mechanism of the chemoprevention by β-caryophyllene. Although further studies are required to confirm the in vivo efficacy of β-caryophyllene, the present results suggest a novel strategy to reduce the harmful effect of smoke in cancer patients and to improve the survival expectations in breast cancer women.
Keywords: IL-8; STAT3; autophagy; breast cancer; caryophyllene sesquiterpenes; cell migration; chemoresistance; cigarette smoke; environmental pollution; γH2AX.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
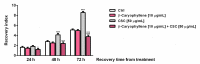



Similar articles
-
Caryophyllane sesquiterpenes inhibit DNA-damage by tobacco smoke in bacterial and mammalian cells.Food Chem Toxicol. 2018 Jan;111:393-404. doi: 10.1016/j.fct.2017.11.018. Epub 2017 Nov 14. Food Chem Toxicol. 2018. PMID: 29154797
-
Modulation of STAT3 Signaling, Cell Redox Defenses and Cell Cycle Checkpoints by β-Caryophyllene in Cholangiocarcinoma Cells: Possible Mechanisms Accounting for Doxorubicin Chemosensitization and Chemoprevention.Cells. 2020 Apr 2;9(4):858. doi: 10.3390/cells9040858. Cells. 2020. PMID: 32252311 Free PMC article.
-
Src/STAT3-dependent heme oxygenase-1 induction mediates chemoresistance of breast cancer cells to doxorubicin by promoting autophagy.Cancer Sci. 2015 Aug;106(8):1023-32. doi: 10.1111/cas.12712. Epub 2015 Jul 4. Cancer Sci. 2015. PMID: 26041409 Free PMC article.
-
Paternal smoking and germ cell death: A mechanistic link to the effects of cigarette smoke on spermatogenesis and possible long-term sequelae in offspring.Mol Cell Endocrinol. 2016 Nov 5;435:85-93. doi: 10.1016/j.mce.2016.07.015. Epub 2016 Jul 14. Mol Cell Endocrinol. 2016. PMID: 27424142 Free PMC article. Review.
-
β-caryophyllene and β-caryophyllene oxide-natural compounds of anticancer and analgesic properties.Cancer Med. 2016 Oct;5(10):3007-3017. doi: 10.1002/cam4.816. Epub 2016 Sep 30. Cancer Med. 2016. PMID: 27696789 Free PMC article. Review.
Cited by
-
Exploring β-caryophyllene: a non-psychotropic cannabinoid's potential in mitigating cognitive impairment induced by sleep deprivation.Arch Pharm Res. 2025 Jan;48(1):1-42. doi: 10.1007/s12272-024-01523-z. Epub 2024 Dec 9. Arch Pharm Res. 2025. PMID: 39653971 Review.
-
Characterization of the Chemopreventive Properties of Cannabis sativa L. Inflorescences from Monoecious Cultivars Grown in Central Italy.Plants (Basel). 2023 Nov 9;12(22):3814. doi: 10.3390/plants12223814. Plants (Basel). 2023. PMID: 38005711 Free PMC article.
-
The Potential Therapeutic Role of Beta-Caryophyllene as a Chemosensitizer and an Inhibitor of Angiogenesis in Cancer.Molecules. 2025 Apr 14;30(8):1751. doi: 10.3390/molecules30081751. Molecules. 2025. PMID: 40333803 Free PMC article. Review.
-
STAT3 Pathways Contribute to β-HCH Interference with Anticancer Tyrosine Kinase Inhibitors.Int J Mol Sci. 2024 Jun 4;25(11):6181. doi: 10.3390/ijms25116181. Int J Mol Sci. 2024. PMID: 38892372 Free PMC article.
-
Essential Oil of Bursera morelensis Promotes Cell Migration on Fibroblasts: In Vitro Assays.Molecules. 2023 Aug 26;28(17):6258. doi: 10.3390/molecules28176258. Molecules. 2023. PMID: 37687087 Free PMC article.
References
-
- Mladěnka P., Applová L., Patočka J., Costa V.M., Remiao F., Pourová J., Mladěnka A., Karlíčková J., Jahodář L., Vopršalová M., et al. Tox-oer and Cardiotox Hradec Králové researchers and collaborators. Comprehensive review of cardiovascular toxicity of drugs and related agents. Med. Res. Rev. 2018;38:1332–1403. doi: 10.1002/med.21476. - DOI - PMC - PubMed
-
- Turner M.C., Andersen Z.J., Baccarelli A., Diver W.R., Gapstur S.M., Pope C.A., Prada D., Samet J., Thurston G., Cohen A. Outdoor air pollution and cancer: An overview of the current evidence and public health recommendations. CA Cancer J. Clin. 2020;70:460–479. doi: 10.3322/caac.21632. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

