Effect of cx-DHED on Abnormal Glucose Transporter Expression Induced by AD Pathologies in the 5xFAD Mouse Model
- PMID: 36142509
- PMCID: PMC9505457
- DOI: 10.3390/ijms231810602
Effect of cx-DHED on Abnormal Glucose Transporter Expression Induced by AD Pathologies in the 5xFAD Mouse Model
Abstract
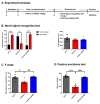
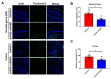
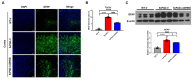
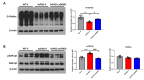
Alzheimer's disease (AD) is a form of dementia associated with abnormal glucose metabolism resulting from amyloid-beta (Aβ) plaques and intracellular neurofibrillary tau protein tangles. In a previous study, we confirmed that carboxy-dehydroevodiamine∙HCl (cx-DHED), a derivative of DHED, was effective at improving cognitive impairment and reducing phosphorylated tau levels and synaptic loss in an AD mouse model. However, the specific mechanism of action of cx-DHED is unclear. In this study, we investigated how the cx-DHED attenuates AD pathologies in the 5xFAD mouse model, focusing particularly on abnormal glucose metabolism. We analyzed behavioral changes and AD pathologies in mice after intraperitoneal injection of cx-DHED for 2 months. As expected, cx-DHED reversed memory impairment and reduced Aβ plaques and astrocyte overexpression in the brains of 5xFAD mice. Interestingly, cx-DHED reversed the abnormal expression of glucose transporters in the brains of 5xFAD mice. In addition, otherwise low O-GlcNac levels increased, and the overactivity of phosphorylated GSK-3β decreased in the brains of cx-DHED-treated 5xFAD mice. Finally, the reduction in synaptic proteins was found to also improve by treatment with cx-DHED. Therefore, we specifically demonstrated the protective effects of cx-DHED against AD pathologies and suggest that cx-DHED may be a potential therapeutic drug for AD.
Keywords: Alzheimer’s disease; GSK-3β; O-GlcNac; cx-DHED; glucose transport.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the study design; collection, analyses, or interpretation of data; writing of the manuscript; or decision to publish the results.
Figures



Similar articles
-
Effects of a Dehydroevodiamine-Derivative on Synaptic Destabilization and Memory Impairment in the 5xFAD, Alzheimer's Disease Mouse Model.Front Behav Neurosci. 2018 Nov 13;12:273. doi: 10.3389/fnbeh.2018.00273. eCollection 2018. Front Behav Neurosci. 2018. PMID: 30483077 Free PMC article.
-
TrkB reduction exacerbates Alzheimer's disease-like signaling aberrations and memory deficits without affecting β-amyloidosis in 5XFAD mice.Transl Psychiatry. 2015 May 5;5(5):e562. doi: 10.1038/tp.2015.55. Transl Psychiatry. 2015. PMID: 25942043 Free PMC article.
-
Butyrylcholinesterase-knockout reduces fibrillar β-amyloid and conserves 18FDG retention in 5XFAD mouse model of Alzheimer's disease.Brain Res. 2017 Sep 15;1671:102-110. doi: 10.1016/j.brainres.2017.07.009. Epub 2017 Jul 17. Brain Res. 2017. PMID: 28729192
-
Diabetes mellitus and Alzheimer's disease: GSK-3β as a potential link.Behav Brain Res. 2018 Feb 26;339:57-65. doi: 10.1016/j.bbr.2017.11.015. Epub 2017 Nov 21. Behav Brain Res. 2018. PMID: 29158110 Review.
-
Synaptic Mitochondria: An Early Target of Amyloid-β and Tau in Alzheimer's Disease.J Alzheimers Dis. 2021;84(4):1391-1414. doi: 10.3233/JAD-215139. J Alzheimers Dis. 2021. PMID: 34719499 Review.
Cited by
-
Liuwei Dihuang formula ameliorates chronic stress-induced emotional and cognitive impairments in mice by elevating hippocampal O-GlcNAc modification.Front Neurosci. 2023 Apr 20;17:1134176. doi: 10.3389/fnins.2023.1134176. eCollection 2023. Front Neurosci. 2023. PMID: 37152609 Free PMC article.
-
Axonal energy metabolism, and the effects in aging and neurodegenerative diseases.Mol Neurodegener. 2023 Jul 20;18(1):49. doi: 10.1186/s13024-023-00634-3. Mol Neurodegener. 2023. PMID: 37475056 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

