Composition of Maternal Circulating Short-Chain Fatty Acids in Gestational Diabetes Mellitus and Their Associations with Placental Metabolism
- PMID: 36145103
- PMCID: PMC9505713
- DOI: 10.3390/nu14183727
Composition of Maternal Circulating Short-Chain Fatty Acids in Gestational Diabetes Mellitus and Their Associations with Placental Metabolism
Abstract
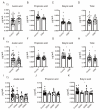
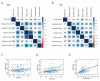
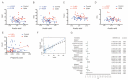
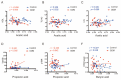
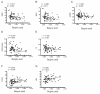
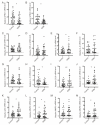
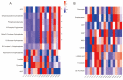
Short-chain fatty acids (SCFAs), which are produced by gut microbiota from dietary fiber, have become candidates for gestational diabetes mellitus (GDM) treatment. However, the associations of circulating SCFAs with maternal-neonatal clinical parameters in GDM and further influences on placental immune-metabolic responses are unclear. Acetate, propionate, and butyrate were decreased in GDM during the second and third trimesters, especially in those with abnormal glucose tolerance at three "oral glucose tolerance test" time points. Butyrate was closely associated with acetate and propionate in correlation and dynamic trajectory analysis. Moreover, butyrate was negatively correlated with white blood cell counts, neutrophil counts, prepregnancy BMI, gestational weight gain per week before GDM diagnosis, and ponderal index but positively correlated with total cholesterol and low-density lipoprotein levels in all pregnancies. On the premise of reduced SCFA contents in GDM, the placental G-protein-coupled receptors 41 and 43 (GPR41/43) were decreased, and histone deacetylases (HDACs) were increased, accompanied by enhanced inflammatory responses. The metabolic status was disturbed, as evidenced by activated glycolysis in GDM. Maternal circulating acetate, propionate, and butyrate levels were associated with demographic factors in normal and GDM women. They influenced placental function and fetal development at birth through GPRs or HDACs, providing more evidence of their therapeutic capacity for GDM pregnancies.
Keywords: gestational diabetes mellitus (GDM); immune; metabolism; placental metabolome; short-chain fatty acid (SCFAs).
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Short-chain fatty acids as novel therapeutics for gestational diabetes.J Mol Endocrinol. 2020 Aug;65(2):21-34. doi: 10.1530/JME-20-0094. J Mol Endocrinol. 2020. PMID: 32580157
-
Dietary Fibre Modulates Gut Microbiota in Late Pregnancy Without Altering SCFA Levels, and Propionate Treatement Has No Effect on Placental Explant Function.Nutrients. 2025 Apr 1;17(7):1234. doi: 10.3390/nu17071234. Nutrients. 2025. PMID: 40218992 Free PMC article.
-
Obesity and gestational diabetes independently and collectively induce specific effects on placental structure, inflammation and endocrine function in a cohort of South African women.J Physiol. 2023 Apr;601(7):1287-1306. doi: 10.1113/JP284139. Epub 2023 Feb 27. J Physiol. 2023. PMID: 36849131
-
Benefits of short-chain fatty acids and their receptors in inflammation and carcinogenesis.Pharmacol Ther. 2016 Aug;164:144-51. doi: 10.1016/j.pharmthera.2016.04.007. Epub 2016 Apr 23. Pharmacol Ther. 2016. PMID: 27113407 Free PMC article. Review.
-
Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation.Nutrients. 2015 Apr 14;7(4):2839-49. doi: 10.3390/nu7042839. Nutrients. 2015. PMID: 25875123 Free PMC article. Review.
Cited by
-
Oral probiotics increased the proportion of Treg, Tfr, and Breg cells to inhibit the inflammatory response and impede gestational diabetes mellitus.Mol Med. 2023 Sep 8;29(1):122. doi: 10.1186/s10020-023-00716-4. Mol Med. 2023. PMID: 37684563 Free PMC article.
-
Sardinian Infants of Diabetic Mothers: A Metabolomics Observational Study.Int J Mol Sci. 2023 Sep 6;24(18):13724. doi: 10.3390/ijms241813724. Int J Mol Sci. 2023. PMID: 37762025 Free PMC article.
-
The maternal gut microbiome in pregnancy: implications for the developing immune system.Nat Rev Gastroenterol Hepatol. 2024 Jan;21(1):35-45. doi: 10.1038/s41575-023-00864-2. Epub 2023 Dec 14. Nat Rev Gastroenterol Hepatol. 2024. PMID: 38097774 Review.
-
Gut Microbiota and Critical Metabolites: Potential Target in Preventing Gestational Diabetes Mellitus?Microorganisms. 2023 Jun 30;11(7):1725. doi: 10.3390/microorganisms11071725. Microorganisms. 2023. PMID: 37512897 Free PMC article. Review.
-
Butyrate and iso-butyrate: a new perspective on nutrition prevention of gestational diabetes mellitus.Nutr Diabetes. 2024 Apr 25;14(1):24. doi: 10.1038/s41387-024-00276-4. Nutr Diabetes. 2024. PMID: 38658555 Free PMC article.
References
-
- Metzger B.E., Lowe L.P., Dyer A.R., Trimble E.R., Chaovarindr U., Coustan D.R., Hadden D.R., Hadden D.R., McCance D.R., Hod M., et al. Hyperglycemia and adverse pregnancy outcomes. N. Engl. J. Med. 2008;358:1991–2002. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

