Two-in-One Nanoparticle Formulation to Deliver a Tyrosine Kinase Inhibitor and microRNA for Targeting Metabolic Reprogramming and Mitochondrial Dysfunction in Gastric Cancer
- PMID: 36145507
- PMCID: PMC9504622
- DOI: 10.3390/pharmaceutics14091759
Two-in-One Nanoparticle Formulation to Deliver a Tyrosine Kinase Inhibitor and microRNA for Targeting Metabolic Reprogramming and Mitochondrial Dysfunction in Gastric Cancer
Abstract
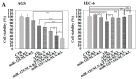
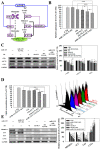
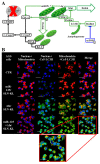
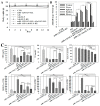
Dysregulational EGFR, KRAS, and mTOR pathways cause metabolic reprogramming, leading to progression of gastric cancer. Afatinib (Afa) is a broad-spectrum tyrosine kinase inhibitor that reduces cancer growth by blocking the EGFR family. MicroRNA 125 (miR-125) reportedly diminishes EGFRs, glycolysis, and anti-apoptosis. Here, a one-shot formulation of miR-125 and Afa was presented for the first time. The formulation comprised solid lipid nanoparticles modified with mitochondrial targeting peptide and EGFR-directed ligand to suppress pan-ErbB-facilitated epithelial-mesenchymal transition and mTOR-mediated metabolism discoordination of glycolysis-glutaminolysis-lipids. Results showed that this cotreatment modulated numerous critical proteins, such as EGFR/HER2/HER3, Kras/ERK/Vimentin, and mTOR/HIF1-α/HK2/LDHA pathways of gastric adenocarcinoma AGS cells. The combinatorial therapy suppressed glutaminolysis, glycolysis, mitochondrial oxidative phosphorylation, and fatty acid synthesis. The cotreatment also notably decreased the levels of lactate, acetyl-CoA, and ATP. The active involvement of mitophagy supported the direction of promoting the apoptosis of AGS cells, which subsequently caused the breakdown of tumor-cell homeostasis and death. In vivo findings in AGS-bearing mice confirmed the superiority of the anti-tumor efficacy and safety of this combination nanomedicine over other formulations. This one-shot formulation disturbed the metabolic reprogramming; alleviated the "Warburg effect" of tumors; interrupted the supply of fatty acid, cholesterol, and triglyceride; and exacerbated the energy depletion in the tumor microenvironment, thereby inhibiting tumor proliferation and aggressiveness. Collectively, the results showed that the two-in-one nanoparticle formulation of miR-125 and Afa was a breakthrough in simplifying drug preparation and administration, as well as effectively inhibiting tumor progression through the versatile targeting of pan-ErbB- and mTOR-mediated mitochondrial dysfunction and dysregulated metabolism.
Keywords: microRNA; mitochondrial dysfunction; mitochondrial targeting; nanoparticle; tumor metabolism reprogramming; tyrosine kinase inhibitor.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures










Similar articles
-
Improving the anticancer effect of afatinib and microRNA by using lipid polymeric nanoparticles conjugated with dual pH-responsive and targeting peptides.J Nanobiotechnology. 2019 Aug 19;17(1):89. doi: 10.1186/s12951-019-0519-6. J Nanobiotechnology. 2019. PMID: 31426807 Free PMC article.
-
Mitochondrion-Directed Nanoparticles Loaded with a Natural Compound and a microRNA for Promoting Cancer Cell Death via the Modulation of Tumor Metabolism and Mitochondrial Dynamics.Pharmaceutics. 2020 Aug 11;12(8):756. doi: 10.3390/pharmaceutics12080756. Pharmaceutics. 2020. PMID: 32796618 Free PMC article.
-
Danusertib, a potent pan-Aurora kinase and ABL kinase inhibitor, induces cell cycle arrest and programmed cell death and inhibits epithelial to mesenchymal transition involving the PI3K/Akt/mTOR-mediated signaling pathway in human gastric cancer AGS and NCI-N78 cells.Drug Des Devel Ther. 2015 Mar 2;9:1293-318. doi: 10.2147/DDDT.S74964. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 25767376 Free PMC article.
-
Metabolic reprogramming in cancer cells: glycolysis, glutaminolysis, and Bcl-2 proteins as novel therapeutic targets for cancer.World J Surg Oncol. 2016 Jan 20;14(1):15. doi: 10.1186/s12957-016-0769-9. World J Surg Oncol. 2016. PMID: 26791262 Free PMC article. Review.
-
Metabolic dysregulation and emerging therapeutical targets for hepatocellular carcinoma.Acta Pharm Sin B. 2022 Feb;12(2):558-580. doi: 10.1016/j.apsb.2021.09.019. Epub 2021 Sep 25. Acta Pharm Sin B. 2022. PMID: 35256934 Free PMC article. Review.
Cited by
-
Global Research Trends and Hotspots in the Role of Cholesterol in Colorectal Cancer: A Bibliometric Analysis.Hum Mutat. 2025 May 24;2025:6546114. doi: 10.1155/humu/6546114. eCollection 2025. Hum Mutat. 2025. PMID: 40452858 Free PMC article.
References
-
- Ebert K., Zwingenberger G., Barbaria E., Keller S., Heck C., Arnold R., Hollerieth V., Mattes J., Geffers R., Raimúndez E., et al. Determining the effects of trastuzumab, cetuximab and afatinib by phosphoprotein, gene expression and phenotypic analysis in gastric cancer cell lines. BMC Cancer. 2020;20:1039. - PMC - PubMed
-
- Corso S., Pietrantonio F., Apicella M., Migliore C., Conticelli D., Petrelli A., D’Errico L., Durando S., Moya-Rull D., Bellomo S.E., et al. Optimized EGFR blockade strategies in EGFR addicted gastroesophageal adenocarcinomas. Clin. Cancer Res. 2021;27:3126–3140. doi: 10.1158/1078-0432.CCR-20-0121. - DOI - PubMed
-
- Chen Z., Liu Z., Zhang M., Huang W., Li Z., Wang S., Zhang C., Dong B., Gao J., Shen L. EPHA2 blockade reverses acquired resistance to afatinib induced by EPHA2-mediated MAPK pathway activation in gastric cancer cells and avatar mice. Int. J. Cancer. 2019;145:2440–2449. doi: 10.1002/ijc.32313. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

