Peg-Grafted Liposomes for L-Asparaginase Encapsulation
- PMID: 36145567
- PMCID: PMC9503594
- DOI: 10.3390/pharmaceutics14091819
Peg-Grafted Liposomes for L-Asparaginase Encapsulation
Abstract
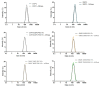
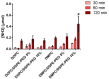
L-asparaginase (ASNase) is an important biological drug used to treat Acute Lymphoblastic Leukemia (ALL). It catalyzes the hydrolysis of L-asparagine (Asn) in the bloodstream and, since ALL cells cannot synthesize Asn, protein synthesis is impaired leading to apoptosis. Despite its therapeutic importance, ASNase treatment is associated to side effects, mainly hypersensitivity and immunogenicity. Furthermore, degradation by plasma proteases and immunogenicity shortens the enzyme half-life. Encapsulation of ASNase in liposomes, nanostructures formed by the self-aggregation of phospholipids, is an attractive alternative to protect the enzyme from plasma proteases and enhance pharmacokinetics profile. In addition, PEGylation might prolong the in vivo circulation of liposomes owing to the spherical shielding conferred by the polyethylene (PEG) corona around the nanostructures. In this paper, ASNase was encapsulated in liposomal formulations composed by 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) or 1,2-dimyristoyl-sn-glycero-3-phosphocholine (DMPC) containing or not different concentrations of 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N [methoxy (polyethylene glycol)-2000] (DSPE-PEG). Nanostructures of approximately 142-202 nm of diameter and polydispersity index (PDI) of 0.069 to 0.190 were obtained and the vesicular shape confirmed by Transmission Electron Microscopy (TEM and cryo-TEM). The encapsulation efficiency (%EE) varied from 10% to 16%. All formulations presented activity in contact with ASNase substrate, indicating the liposomes permeability to Asn and/or enzyme adsorption at the nanostructures' surface; the highest activity was observed for DMPC/DSPE-PEG 10%. Finally, we investigated the activity against the Molt 4 leukemic cell line and found a lower IC50 for the DMPC/DSPE-PEG 10% formulation in comparison to the free enzyme, indicating our system could provide in vivo activity while protecting the enzyme from immune system recognition and proteases degradation.
Keywords: L-asparaginase; acute lymphoblastic leukemia; liposome; nanocarrier; nanoreactor; pegylated liposome.
Conflict of interest statement
The authors declare no conflict of interest, financial or otherwise.
Figures









References
-
- Santos J.H.P.M., Costa I.M., Molino J.V.D., Leite M.S.M., Pimenta M.V., Coutinho J.A.P., Pessoa A., Ventura S.P.M., Lopes A.M., Monteiro G. Heterologous Expression and Purification of Active L-Asparaginase I of Saccharomyces Cerevisiae in Escherichia Coli Host. Biotechnol. Prog. 2017;33:416–424. doi: 10.1002/btpr.2410. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

