Auxotrophic Lactobacillus Expressing Porcine Rotavirus VP4 Constructed Using CRISPR-Cas9D10A System Induces Effective Immunity in Mice
- PMID: 36146587
- PMCID: PMC9504633
- DOI: 10.3390/vaccines10091510
Auxotrophic Lactobacillus Expressing Porcine Rotavirus VP4 Constructed Using CRISPR-Cas9D10A System Induces Effective Immunity in Mice
Abstract
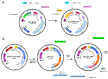
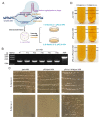
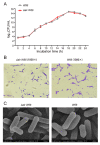
Porcine rotavirus (PoRV) mainly causes acute diarrhea in piglets under eight weeks of age and has potentially high morbidity and mortality rates. As vaccine carriers for oral immunization, lactic acid bacteria (LAB) are an ideal strategy for blocking PoRV infections. However, the difficulty in knocking out specific genes, inserting foreign genes, and the residues of antibiotic selection markers are major challenges for the oral vaccination of LAB. In this study, the target gene, alanine racemase (alr), in the genome of Lactobacillus casei strain W56 (L. casei W56) was knocked out to construct an auxotrophic L. casei strain (L. casei Δalr W56) using the CRISPR-Cas9D10A gene editing system. A recombinant strain (pPG-alr-VP4/Δalr W56) was constructed using an electrotransformed complementary plasmid. Expression of the alr-VP4 fusion protein from pPG-alr-VP4/Δalr W56 was detected using Western blotting. Mice orally immunized with pPG-alr-VP4/Δalr W56 exhibited high levels of serum IgG and mucosal secretory immunoglobulin A (SIgA), which exhibited neutralizing effects against PoRV. Cytokines levels in serum detected using ELISA, indicated that the recombinant strain induced an immune response dominated by Th2 cells. Our data suggest that pPG-alr-VP4/Δalr W56, an antibiotic-resistance-free LAB, provides a safer vaccine strategy against PoRV infection.
Keywords: CRISPR/Cas9D10A; Lactobacillus; nutritional deficient; oral immunization; porcine rotaviruses.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Evaluation of the immunogenicity of auxotrophic Lactobacillus with CRISPR-Cas9D10A system-mediated chromosomal editing to express porcine rotavirus capsid protein VP4.Virulence. 2022 Dec;13(1):1315-1330. doi: 10.1080/21505594.2022.2107646. Virulence. 2022. PMID: 35920261 Free PMC article.
-
Immunogenicity of Recombinant-Deficient Lactobacillus casei with Complementary Plasmid Expressing Alanine Racemase Gene and Core Neutralizing Epitope Antigen against Porcine Epidemic Diarrhea Virus.Vaccines (Basel). 2021 Sep 26;9(10):1084. doi: 10.3390/vaccines9101084. Vaccines (Basel). 2021. PMID: 34696192 Free PMC article.
-
Recombinant porcine rotavirus VP4 and VP4-LTB expressed in Lactobacillus casei induced mucosal and systemic antibody responses in mice.BMC Microbiol. 2009 Dec 4;9:249. doi: 10.1186/1471-2180-9-249. BMC Microbiol. 2009. PMID: 19958557 Free PMC article.
-
Lactiplantibacillus plantarum surface-displayed VP6 (PoRV) protein can prevent PoRV infection in piglets.Int Immunopharmacol. 2024 May 30;133:112079. doi: 10.1016/j.intimp.2024.112079. Epub 2024 Apr 13. Int Immunopharmacol. 2024. PMID: 38615376
-
Oral recombinant Lactobacillus vaccine targeting the intestinal microfold cells and dendritic cells for delivering the core neutralizing epitope of porcine epidemic diarrhea virus.Microb Cell Fact. 2018 Feb 9;17(1):20. doi: 10.1186/s12934-018-0861-7. Microb Cell Fact. 2018. PMID: 29426335 Free PMC article.
Cited by
-
Design of multi-epitope vaccine against porcine rotavirus using computational biology and molecular dynamics simulation approaches.Virol J. 2024 Jul 22;21(1):160. doi: 10.1186/s12985-024-02440-9. Virol J. 2024. PMID: 39039549 Free PMC article.
-
Assessing immunogenicity of CRISPR-NCas9 engineered strain against porcine epidemic diarrhea virus.Appl Microbiol Biotechnol. 2024 Mar 2;108(1):248. doi: 10.1007/s00253-023-12989-0. Appl Microbiol Biotechnol. 2024. PMID: 38430229 Free PMC article.
-
Portulaca oleracea L. Polysaccharide Inhibits Porcine Rotavirus In Vitro.Animals (Basel). 2023 Jul 14;13(14):2306. doi: 10.3390/ani13142306. Animals (Basel). 2023. PMID: 37508085 Free PMC article.
-
Lactobacillus acidophilus Expressing Murine Rotavirus VP8 and Mucosal Adjuvants Induce Virus-Specific Immune Responses.Vaccines (Basel). 2023 Nov 28;11(12):1774. doi: 10.3390/vaccines11121774. Vaccines (Basel). 2023. PMID: 38140179 Free PMC article.
-
Advancing vaccine technology through the manipulation of pathogenic and commensal bacteria.Mater Today Bio. 2024 Nov 16;29:101349. doi: 10.1016/j.mtbio.2024.101349. eCollection 2024 Dec. Mater Today Bio. 2024. PMID: 39850273 Free PMC article. Review.
References
-
- Li Y.J., Ma G.P., Li G.W., Qiao X.Y., Ge J.W., Tang L.J., Liu M., Liu L.W. Oral vaccination with the porcine rotavirus VP4 outer capsid protein expressed by Lactococcus lactis induces specific antibody production. J. Biomed. Biotechnol. 2010;2010:708460. doi: 10.1155/2010/708460. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

