Double and Triple Combinations of Broadly Neutralizing Antibodies Provide Efficient Neutralization of All HIV-1 Strains from the Global Panel
- PMID: 36146719
- PMCID: PMC9503787
- DOI: 10.3390/v14091910
Double and Triple Combinations of Broadly Neutralizing Antibodies Provide Efficient Neutralization of All HIV-1 Strains from the Global Panel
Abstract
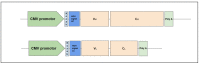
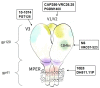
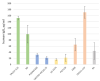
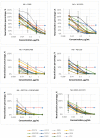
The use of broadly neutralizing antibodies (bNAbs) is a promising approach to HIV-1 treatment. In this work, we evaluate the neutralizing activity of the following HIV-1 bNAbs: VCR07-523, N6, PGDM1400, CAP256-VRC26.25, 10-1074, PGT128, 10E8, and DH511.11P, which are directed to different Env surface epitopes. We used the global panel of HIV-1 pseudoviruses to analyze the bNAbs' potency and chose the most potent ones. To achieve maximum neutralization breadth and minimum IC50 concentration, the most effective antibodies were tested in double and triple combinations. Among the doubles, the combinations of N6+PGDM1400 and N6+PGT128 with IC50 ≤ 0.3 µg/mL proved to be the most effective. The most effective triple combination was N6+PGDM1400+PGT128. Our data demonstrate that this combination neutralizes pseudoviruses of the global HIV-1 panel with IC50 ≤ 0.11 µg/mL and IC80 ≤ 0.25 µg/mL.
Keywords: 10-1074; 10E8; CAP256-VRC26.25; DH511.11P; Env; HIV-1; HIV-1 global panel; N6; PGDM1400; PGT128; VCR07-523; broadly neutralizing antibodies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
Triple Combinations of AAV9-Vectors Encoding Anti-HIV bNAbs Provide Long-Term In Vivo Expression of Human IgG Effectively Neutralizing Pseudoviruses from HIV-1 Global Panel.Viruses. 2024 Aug 14;16(8):1296. doi: 10.3390/v16081296. Viruses. 2024. PMID: 39205270 Free PMC article.
-
Neutralization diversity of HIV-1 Indian subtype C envelopes obtained from cross sectional and followed up individuals against broadly neutralizing monoclonal antibodies having distinct gp120 specificities.Retrovirology. 2021 May 14;18(1):12. doi: 10.1186/s12977-021-00556-2. Retrovirology. 2021. PMID: 33990195 Free PMC article.
-
Subtle Longitudinal Alterations in Env Sequence Potentiate Differences in Sensitivity to Broadly Neutralizing Antibodies following Acute HIV-1 Subtype C Infection.J Virol. 2022 Dec 21;96(24):e0127022. doi: 10.1128/jvi.01270-22. Epub 2022 Dec 1. J Virol. 2022. PMID: 36453881 Free PMC article.
-
Polyfunctionality of broadly neutralizing HIV-1 antibodies.Curr Opin HIV AIDS. 2023 Jul 1;18(4):178-183. doi: 10.1097/COH.0000000000000799. Epub 2023 May 9. Curr Opin HIV AIDS. 2023. PMID: 37249912 Review.
-
Broadly neutralizing antibodies for treatment and prevention of HIV-1 infection.Curr Opin HIV AIDS. 2022 Jul 1;17(4):247-257. doi: 10.1097/COH.0000000000000742. Curr Opin HIV AIDS. 2022. PMID: 35762380 Review.
Cited by
-
Recapitulation of HIV-1 Neutralization Breadth in Plasma by the Combination of Two Broadly Neutralizing Antibodies from Different Lineages in the Same SHIV-Infected Rhesus Macaque.Int J Mol Sci. 2024 Jun 29;25(13):7200. doi: 10.3390/ijms25137200. Int J Mol Sci. 2024. PMID: 39000308 Free PMC article.
-
Broadly neutralizing antibodies for HIV prevention: a comprehensive review and future perspectives.Clin Microbiol Rev. 2024 Jun 13;37(2):e0015222. doi: 10.1128/cmr.00152-22. Epub 2024 Apr 30. Clin Microbiol Rev. 2024. PMID: 38687039 Free PMC article. Review.
-
Triple Combinations of AAV9-Vectors Encoding Anti-HIV bNAbs Provide Long-Term In Vivo Expression of Human IgG Effectively Neutralizing Pseudoviruses from HIV-1 Global Panel.Viruses. 2024 Aug 14;16(8):1296. doi: 10.3390/v16081296. Viruses. 2024. PMID: 39205270 Free PMC article.
References
-
- UNAIDS AIDS Statistics. [(accessed on 25 April 2022)]. Available online: https://www.unaids.org/ru/resources/fact-sheet.
-
- WHO. [(accessed on 25 April 2022)]. Available online: https://www.who.int/news-room/fact-sheets/detail/hiv-drugresistance.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

