miR-182-5p attenuates Schistosoma japonicum-induced hepatic fibrosis by targeting tristetraprolin
- PMID: 36148947
- PMCID: PMC9828319
- DOI: 10.3724/abbs.2022130
miR-182-5p attenuates Schistosoma japonicum-induced hepatic fibrosis by targeting tristetraprolin
Abstract
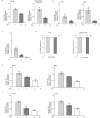
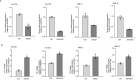
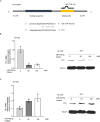
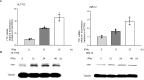
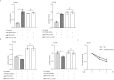
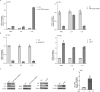
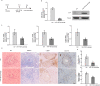
Egg granuloma formation in the liver is the main pathological lesion caused by Schistosoma japonicum infection, which generally results in liver fibrosis and may lead to death in advanced patients. MicroRNAs (miRNAs) regulate the process of liver fibrosis, but the putative function of miRNAs in liver fibrosis induced by S. japonicum infection is largely unclear. Here, we detect a new miRNA, miR-182-5p, which shows significantly decreased expression in mouse livers after stimulation by soluble egg antigen (SEA) of S. japonicum or S. japonicum infection. Knockdown or overexpression of miR-182-5p in vitro causes the increased or decreased expression of tristetraprolin (TTP), an important immunosuppressive protein in the process of liver fibrosis. Furthermore, knockdown of miR-182-5p in vivo upregulates TTP expression and significantly alleviates S. japonicum-induced hepatic fibrosis. Our data demonstrate that downregulation of miR-182-5p increases the expression of TTP in mouse livers following schistosome infection, which leads to destabilization of inflammatory factor mRNAs and attenuates liver fibrosis. Our results uncover fine-tuning of liver inflammatory reactions related to liver fibrosis caused by S. japonicum infection and provide new insights into the regulation of schistosomiasis-induced hepatic fibrosis.
Keywords: liver fibrosis; miR-182-5p; microRNA; tristetraprolin.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
Similar articles
-
MiR-383-5p promotes schistosomiasis-induced liver fibrosis by targeting peroxiredoxin-3.Parasit Vectors. 2025 Jun 3;18(1):205. doi: 10.1186/s13071-025-06824-w. Parasit Vectors. 2025. PMID: 40462224 Free PMC article.
-
MicroRNA-29a-3p prevents Schistosoma japonicum-induced liver fibrosis by targeting Roundabout homolog 1 in hepatic stellate cells.Parasit Vectors. 2023 Jun 6;16(1):184. doi: 10.1186/s13071-023-05791-4. Parasit Vectors. 2023. PMID: 37280619 Free PMC article.
-
Upregulation of KSRP by miR-27b attenuates schistosomiasis-induced hepatic fibrosis by targeting TGF-β1.FASEB J. 2020 Mar;34(3):4120-4133. doi: 10.1096/fj.201902438R. Epub 2020 Jan 17. FASEB J. 2020. PMID: 31953889
-
MicroRNA dysregulation in schistosomiasis-induced hepatic fibrosis: a systematic review.Expert Rev Mol Diagn. 2023 Mar;23(3):257-265. doi: 10.1080/14737159.2023.2182190. Epub 2023 Feb 26. Expert Rev Mol Diagn. 2023. PMID: 36803616
-
Pathology and molecular mechanisms of Schistosoma japonicum-associated liver fibrosis.Front Cell Infect Microbiol. 2022 Oct 28;12:1035765. doi: 10.3389/fcimb.2022.1035765. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36389166 Free PMC article. Review.
Cited by
-
Tristetraprolin Family Members and Processing Bodies: A Complex Regulatory Network Involved in Fatty Liver Disease, Viral Hepatitis and Hepatocellular Carcinoma.Cancers (Basel). 2025 Jan 21;17(3):348. doi: 10.3390/cancers17030348. Cancers (Basel). 2025. PMID: 39941720 Free PMC article. Review.
-
MicroRNAs in infectious diseases: potential diagnostic biomarkers and therapeutic targets.Clin Microbiol Rev. 2023 Dec 20;36(4):e0001523. doi: 10.1128/cmr.00015-23. Epub 2023 Nov 1. Clin Microbiol Rev. 2023. PMID: 37909789 Free PMC article. Review.
References
-
- WHO. Schistosomiasis. https://www.who.int/schistosomiasis/en/.2022.
-
- LoVerde PT. Schistosomiasis. Adv Exp Med Biol 2019. , 1154: 45–70 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

