Supplementation with uric and ascorbic acid protects stored red blood cells through enhancement of non-enzymatic antioxidant activity and metabolic rewiring
- PMID: 36155342
- PMCID: PMC9513173
- DOI: 10.1016/j.redox.2022.102477
Supplementation with uric and ascorbic acid protects stored red blood cells through enhancement of non-enzymatic antioxidant activity and metabolic rewiring
Abstract
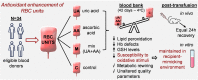
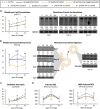
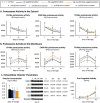
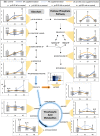
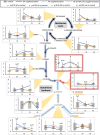
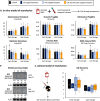
Redox imbalance and oxidative stress have emerged as generative causes of the structural and functional degradation of red blood cells (RBC) that happens during their hypothermic storage at blood banks. The aim of the present study was to examine whether the antioxidant enhancement of stored RBC units following uric (UA) and/or ascorbic acid (AA) supplementation can improve their storability as well as post-transfusion phenotypes and recovery by using in vitro and animal models, respectively. For this purpose, 34 leukoreduced CPD/SAGM RBC units were aseptically split in 4 satellite units each. UA, AA or their mixture were added in the three of them, while the fourth was used as control. Hemolysis as well as redox and metabolic parameters were studied in RBC units throughout storage. The addition of antioxidants maintained the quality parameters of stored RBCs, (e.g., hemolysis, calcium homeostasis) and furthermore, shielded them against oxidative defects by boosting extracellular and intracellular (e.g., reduced glutathione; GSH) antioxidant powers. Higher levels of GSH seemed to be obtained through distinct metabolic rewiring in the modified units: methionine-cysteine metabolism in UA samples and glutamine production in the other two groups. Oxidatively-induced hemolysis, reactive oxygen species accumulation and membrane lipid peroxidation were lower in all modifications compared to controls. Moreover, denatured/oxidized Hb binding to the membrane was minor, especially in the AA and mix treatments during middle storage. The treated RBC were able to cope against pro-oxidant triggers when found in a recipient mimicking environment in vitro, and retain control levels of 24h recovery in mice circulation. The currently presented study provides (a) a detailed picture of the effect of UA/AA administration upon stored RBCs and (b) insight into the differential metabolic rewiring when distinct antioxidant "enhancers" are used.
Keywords: Antioxidant supplementation; Ascorbic acid; Glutathione metabolism; Oxidative stress; RBC storage lesion; Uric acid.
Copyright © 2022 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Bardyn M., Tissot J.D., Prudent M. Oxidative stress and antioxidant defenses during blood processing and storage of erythrocyte concentrates. Transfus. Clin. Biol. : J. Soc. Fr. Transfus. Sanguine. 2018;25(1):96–100. - PubMed
-
- Delobel J., Prudent M., Tissot J.D., Lion N. Proteomics of the red blood cell carbonylome during blood banking of erythrocyte concentrates, Proteomics. Clin. Appl. 2016;10(3):257–266. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

