Small-molecule Wnt inhibitors are a potential novel therapy for intestinal fibrosis in Crohns disease
- PMID: 36156078
- PMCID: PMC9583737
- DOI: 10.1042/CS20210889
Small-molecule Wnt inhibitors are a potential novel therapy for intestinal fibrosis in Crohns disease
Abstract
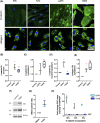
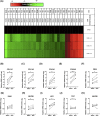
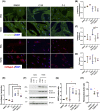
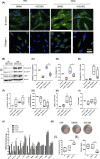
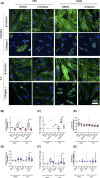
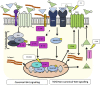
Intestinal fibrosis and stricture formation is an aggressive complication of Crohns disease (CD), linked to increased morbidity and costs. The present study investigates the contribution of Wingless-Int-1 (Wnt) signalling to intestinal fibrogenesis, considers potential cross-talk between Wnt and transforming growth factor β1 (TGFβ) signalling pathways, and assesses the therapeutic potential of small-molecule Wnt inhibitors. β-catenin expression was explored by immunohistochemistry (IHC) in formalin-fixed paraffin embedded (FFPE) tissue from patient-matched nonstrictured (NSCD) and strictured (SCD) intestine (n=6 pairs). Functional interactions between Wnt activation, TGFβ signalling, and type I collagen (Collagen-I) expression were explored in CCD-18Co cells and primary CD myofibroblast cultures established from surgical resection specimens (n=16) using small-molecule Wnt inhibitors and molecular techniques, including siRNA-mediated gene knockdown, immunofluorescence (IF), Wnt gene expression arrays, and western blotting. Fibrotic SCD tissue was marked by an increase in β-catenin-positive cells. In vitro, activation of Wnt-β-catenin signalling increased Collagen-I expression in CCD-18Co cells. Conversely, ICG-001, an inhibitor of β-catenin signalling, reduced Collagen-I expression in cell lines and primary CD myofibroblasts. TGFβ increased β-catenin protein levels but did not activate canonical Wnt signalling. Rather, TGFβ up-regulated WNT5B, a noncanonical Wnt ligand, and the Wnt receptor FZD8, which contributed directly to the up-regulation of Collagen-I through a β-catenin-independent mechanism. Treatment of CCD-18Co fibroblasts and patient-derived myofibroblasts with the FZD8 inhibitor 3235-0367 reduced extracellular matrix (ECM) expression. Our data highlight small-molecule Wnt inhibitors of both canonical and noncanonical Wnt signalling, as potential antifibrotic drugs to treat SCD intestinal fibrosis. They also highlight the importance of the cross-talk between Wnt and TGFβ signalling pathways in CD intestinal fibrosis.
Keywords: Crohns disease; Wnt proteins; fibrosis; inhibitors; intestine.
© 2022 The Author(s).
Conflict of interest statement
The authors declare that there are no competing interests associated with the manuscript.
Figures

References
-
- Murthy S.K., Begum J., Benchimol E.I., Bernstein C.N., Kaplan G.G., McCurdy J.D.et al. . (2020) Introduction of anti-TNF therapy has not yielded expected declines in hospitalisation and intestinal resection rates in inflammatory bowel diseases: a population-based interrupted time series study. Gut 69, 274–282 10.1136/gutjnl-2019-318440 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

