Properties and modulation of excitatory inputs to the locus coeruleus
- PMID: 36156249
- PMCID: PMC9669264
- DOI: 10.1113/JP283605
Properties and modulation of excitatory inputs to the locus coeruleus
Abstract
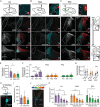
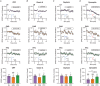
Excitatory inputs drive burst firing of locus coeruleus (LC) noradrenaline (NA) neurons in response to a variety of stimuli. Though a small number of glutamatergic LC afferents have been investigated, the overall landscape of these excitatory inputs is largely unknown. The current study used an optogenetic approach to isolate three glutamatergic afferents: the prefrontal cortex (PFC), lateral hypothalamus (LH) and periaqueductal grey (PAG). AAV5-DIO-ChR2 was injected into each region in male and female CaMKII-Cre mice and the properties of excitatory inputs on LC-NA cells were measured. Notably we found differences among these inputs. First, the pattern of axonal innervation differed between inputs such that LH afferents were concentrated in the posterior portion of the LC-NA somatic region while PFC afferents were denser in the medial dendritic region. Second, basal intrinsic properties varied for afferents, with LH inputs having the highest connectivity and the largest amplitude excitatory postsynaptic currents while PAG inputs had the lowest initial release probability. Third, while orexin and oxytocin had minimal effects on any input, dynorphin strongly inhibited excitatory inputs originating from the LH and PAG, and corticotrophin releasing factor (CRF) selectively inhibited inputs from the PAG. Overall, these results demonstrate that individual afferents to the LC have differing properties, which may contribute to the modularity of the LC and its ability to mediate various behavioural outcomes. KEY POINTS: Excitatory inputs to the locus coeruleus (LC) are important for driving noradrenaline neuron activity and downstream behaviours in response to salient stimuli, but little is known about the functional properties of different glutamate inputs that innervate these neurons We used a virus-mediated optogenetic approach to compare glutamate afferents from the prefrontal cortex (PFC), the lateral hypothalamus (LH) and the periaqueductal grey (PAG). While PFC was predicted to make synaptic inputs, we found that the LH and PAG also drove robust excitatory events in LC noradrenaline neurons. The strength, kinetics, and short-term plasticity of each input differed as did the extent of neuromodulation by both dynorphin and corticotrophin releasing factor. Thus each input displayed a unique set of basal properties and modulation by peptides. This characterization is an important step in deciphering the heterogeneity of the LC.
Keywords: circuit; glutamate; neuromodulation; stress; synaptic.
© 2022 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Figures


Similar articles
-
Corticotropin-releasing factor is preferentially colocalized with excitatory rather than inhibitory amino acids in axon terminals in the peri-locus coeruleus region.Neuroscience. 2001;106(2):375-84. doi: 10.1016/s0306-4522(01)00279-2. Neuroscience. 2001. PMID: 11566507
-
Afferent regulation of locus coeruleus neurons: anatomy, physiology and pharmacology.Prog Brain Res. 1991;88:47-75. doi: 10.1016/s0079-6123(08)63799-1. Prog Brain Res. 1991. PMID: 1687622 Review.
-
Projections from the periaqueductal gray to the rostromedial pericoerulear region and nucleus locus coeruleus: anatomic and physiologic studies.J Comp Neurol. 1991 Apr 15;306(3):480-94. doi: 10.1002/cne.903060311. J Comp Neurol. 1991. PMID: 1713927
-
Regulation of Lateral Hypothalamic Orexin Activity by Local GABAergic Neurons.J Neurosci. 2018 Feb 7;38(6):1588-1599. doi: 10.1523/JNEUROSCI.1925-17.2017. Epub 2018 Jan 8. J Neurosci. 2018. PMID: 29311142 Free PMC article.
-
Convergent regulation of locus coeruleus activity as an adaptive response to stress.Eur J Pharmacol. 2008 Apr 7;583(2-3):194-203. doi: 10.1016/j.ejphar.2007.11.062. Epub 2008 Jan 19. Eur J Pharmacol. 2008. PMID: 18255055 Free PMC article. Review.
Cited by
-
Inputs to the locus coeruleus from the periaqueductal gray and rostroventral medulla shape opioid-mediated descending pain modulation.Sci Adv. 2024 Apr 26;10(17):eadj9581. doi: 10.1126/sciadv.adj9581. Epub 2024 Apr 26. Sci Adv. 2024. PMID: 38669335 Free PMC article.
-
Neonatal testosterone exposure alleviates female-specific severity of formalin-induced inflammatory pain in mice.Front Neural Circuits. 2025 Jul 2;19:1593443. doi: 10.3389/fncir.2025.1593443. eCollection 2025. Front Neural Circuits. 2025. PMID: 40671873 Free PMC article.
-
Heterogeneous pericoerulear neurons tune arousal and exploratory behaviours.Nature. 2025 Jul;643(8071):437-447. doi: 10.1038/s41586-025-08952-w. Epub 2025 May 7. Nature. 2025. PMID: 40335695 Free PMC article.
-
Mu opioid receptors gate the locus coeruleus pain generator.bioRxiv [Preprint]. 2024 Aug 28:2023.10.20.562785. doi: 10.1101/2023.10.20.562785. bioRxiv. 2024. PMID: 37961541 Free PMC article. Preprint.
-
A hypothalamus-brainstem circuit governs the prioritization of safety over essential needs.Nat Neurosci. 2025 Jul;28(7):1473-1485. doi: 10.1038/s41593-025-01975-6. Epub 2025 May 28. Nat Neurosci. 2025. PMID: 40437053 Free PMC article.
References
-
- Abercrombie, E. D. , & Jacobs, B. L. (1988). Systemic naloxone administration potentiates locus coeruleus noradrenergic neuronal activity under stressful but not non‐stressful conditions. Brain Research, 441(1–2), 362–366. - PubMed
-
- Arnsten, A. F. , & Goldman‐Rakic, P. S. (1984). Selective prefrontal cortical projections to the region of the locus coeruleus and raphe nuclei in the rhesus monkey. Brain Research, 306(1–2), 9–18. - PubMed
-
- Aston‐Jones, G. , Ennis, M. , Pieribone, V. A. , Nickell, W. T. , & Shipley, M. T. (1986). The brain nucleus locus coeruleus: Restricted afferent control of a broad efferent network. Science, 234(4777), 734–737. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

