The role of RNA m6A methylation in lipid metabolism
- PMID: 36157445
- PMCID: PMC9492936
- DOI: 10.3389/fendo.2022.866116
The role of RNA m6A methylation in lipid metabolism
Abstract
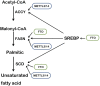
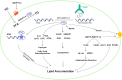
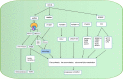
The m6A methylation is the most numerous modification of mRNA in mammals, coordinated by RNA m6A methyltransferases, RNA m6A demethylases, and RNA m6A binding proteins. They change the RNA m6A methylation level in their specific manner. RNA m6A modification has a significant impact on lipid metabolic regulation. The "writer" METTL3/METTL14 and the "eraser" FTO can promote the accumulation of lipids in various cells by affecting the decomposition and synthesis of lipids. The "reader" YTHDF recognizes m6A methylation sites of RNA and regulates the target genes' translation. Due to this function that regulates lipid metabolism, RNA m6A methylation plays a pivotal role in metabolic diseases and makes it a great potential target for therapy.
Keywords: FTO (fat mass and obesity-associated) gene; M6A; METTL3; lipid; obesity.
Copyright © 2022 Wang, Wang, Gu, Su, Gu and Feng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
m6A modification impacts hepatic drug and lipid metabolism properties by regulating carboxylesterase 2.Biochem Pharmacol. 2021 Nov;193:114766. doi: 10.1016/j.bcp.2021.114766. Epub 2021 Sep 16. Biochem Pharmacol. 2021. PMID: 34536357
-
The molecular regulated mechanism of METTL3 and FTO in lipid metabolism of Hu sheep.Genomics. 2024 Nov;116(6):110945. doi: 10.1016/j.ygeno.2024.110945. Epub 2024 Sep 27. Genomics. 2024. PMID: 39341298
-
Upregulation of METTL3 expression and m6A RNA methylation in placental trophoblasts in preeclampsia.Placenta. 2021 Jan 1;103:43-49. doi: 10.1016/j.placenta.2020.10.016. Epub 2020 Oct 13. Placenta. 2021. PMID: 33070036
-
m6A RNA Methylation and Implications for Hepatic Lipid Metabolism.DNA Cell Biol. 2024 Jun;43(6):271-278. doi: 10.1089/dna.2023.0410. Epub 2024 Apr 18. DNA Cell Biol. 2024. PMID: 38635960 Review.
-
RNA m6A modification, signals for degradation or stabilisation?Biochem Soc Trans. 2024 Apr 24;52(2):707-717. doi: 10.1042/BST20230574. Biochem Soc Trans. 2024. PMID: 38629637 Free PMC article. Review.
Cited by
-
The role of the methyltransferase METTL3 in prostate cancer: a potential therapeutic target.BMC Cancer. 2024 Jan 2;24(1):8. doi: 10.1186/s12885-023-11741-1. BMC Cancer. 2024. PMID: 38166703 Free PMC article. Review.
-
In Search of a Function for the N6-Methyladenosine in Epitranscriptome, Autophagy and Neurodegenerative Diseases.Neurol Int. 2023 Aug 10;15(3):967-979. doi: 10.3390/neurolint15030062. Neurol Int. 2023. PMID: 37606395 Free PMC article. Review.
-
N6-Methyladenosine Modification in the Metabolic Dysfunction-Associated Steatotic Liver Disease.Nutrients. 2025 Mar 27;17(7):1158. doi: 10.3390/nu17071158. Nutrients. 2025. PMID: 40218916 Free PMC article. Review.
-
Global m6A methylation and gene expression patterns in human microglial HMC3 cells infected with HIV-1.Heliyon. 2023 Oct 26;9(11):e21307. doi: 10.1016/j.heliyon.2023.e21307. eCollection 2023 Nov. Heliyon. 2023. PMID: 38027859 Free PMC article.
-
Unveiling the Impact of ApoF Deficiency on Liver and Lipid Metabolism: Insights from Transcriptome-Wide m6A Methylome Analysis in Mice.Genes (Basel). 2024 Mar 9;15(3):347. doi: 10.3390/genes15030347. Genes (Basel). 2024. PMID: 38540406 Free PMC article.
References
-
- Cheng L, Zhang S, Shang F, Ning Y, Huang Z, He R, et al. . Emodin improves glucose and lipid metabolism disorders in obese mice via activating brown adipose tissue and inducing browning of white adipose tissue. Front Endocrinol (Lausanne) (2021) 12:618037. doi: 10.3389/fendo.2021.618037 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

