Proteomic Analysis of m.8296A>G Variation in the Mitochondrial tRNA Lys Gene
- PMID: 36158049
- PMCID: PMC9421666
- DOI: 10.1159/000519526
Proteomic Analysis of m.8296A>G Variation in the Mitochondrial tRNA Lys Gene
Abstract
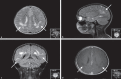
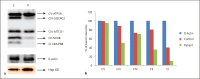
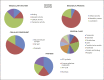
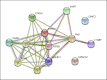
Variation in the mitochondrial tRNA Lys gene at position 8296 was previously found to be associated with maternally inherited diabetes mellitus and deafness, hypertrophic cardiomyopathy, myoclonic epilepsy with ragged-red fibers and mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes. The pathogenicity of the m.8296A>G variation is unclear. In this study, we aimed to analyze the mitochondrial proteome in a patient with m.8296A>G variation to elucidate the effects of this mutation at the protein level. Whole-exome sequencing and mitochondrial genome analysis were performed in a patient with sensorineural hearing impairment, cognitive impairment, leukodystrophy, migraine-like headaches, and gastrointestinal dysmotility. Mitochondrial genome analysis identified a homoplasmic m.8296A>G variation in the mitochondrial tRNA Lys gene in the proband and unaffected mother. Global mitochondrial proteome analysis was carried out in the muscle mitochondria of the index patient and a control subject. Comparative muscle mitochondrial proteome analysis revealed a total of 13 nuclear-encoded mitochondrial proteins differently expressed with respect to the control. Ten of the 13 proteins were downregulated. Most of the proteins were involved in ATP synthesis and Krebs cycle and have strong interactions with each other. We considered the m.8296A>G variation to be pathogenic with variable penetrance for our patient's phenotype, and this variation led to different expressions of nuclear-encoded proteins involved in energy metabolism.
Keywords: A8296G mutation; Leukodystrophy; Mitochondrial proteome; Muscle mitochondria; Sensorineural hearing impairment.
Copyright © 2022 by S. Karger AG, Basel.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures

Similar articles
-
[Mitochondrial encephalopathy, lactic acidosis and stroke-like episodes / myoclonus epilepsy with ragged-red fibers /Leigh overlap syndrome caused by mitochondrial DNA 8344A>G mutation].Beijing Da Xue Xue Bao Yi Xue Ban. 2020 Oct 18;52(5):851-855. doi: 10.19723/j.issn.1671-167X.2020.05.009. Beijing Da Xue Xue Bao Yi Xue Ban. 2020. PMID: 33047718 Free PMC article. Chinese.
-
Isoleucyl-tRNA synthetase levels modulate the penetrance of a homoplasmic m.4277T>C mitochondrial tRNA(Ile) mutation causing hypertrophic cardiomyopathy.Hum Mol Genet. 2012 Jan 1;21(1):85-100. doi: 10.1093/hmg/ddr440. Epub 2011 Sep 26. Hum Mol Genet. 2012. PMID: 21945886
-
The nuclear background influences the penetrance of the near-homoplasmic m.1630 A > G MELAS variant in a symptomatic proband and asymptomatic mother.Mol Genet Metab. 2019 Apr;126(4):429-438. doi: 10.1016/j.ymgme.2019.01.022. Epub 2019 Jan 25. Mol Genet Metab. 2019. PMID: 30709774 Free PMC article.
-
The Mitochondrial tRNASer(UCN) Gene: A Novel m.7484A>G Mutation Associated with Mitochondrial Encephalomyopathy and Literature Review.Life (Basel). 2023 Feb 16;13(2):554. doi: 10.3390/life13020554. Life (Basel). 2023. PMID: 36836911 Free PMC article. Review.
-
The expanding clinical spectrum of mitochondrial diseases.Brain Dev. 1993 Jan-Feb;15(1):1-22. doi: 10.1016/0387-7604(93)90002-p. Brain Dev. 1993. PMID: 8338207 Review.
Cited by
-
Mitochondrial DNA variants in the pathogenesis and metabolic alterations of diabetes mellitus.Mol Genet Metab Rep. 2024 Dec 28;42:101183. doi: 10.1016/j.ymgmr.2024.101183. eCollection 2025 Mar. Mol Genet Metab Rep. 2024. PMID: 39835172 Free PMC article. Review.
-
Energy metabolism disorders in migraine: triggers, pathways, and therapeutic repurposing.Front Neurol. 2025 Apr 2;16:1561000. doi: 10.3389/fneur.2025.1561000. eCollection 2025. Front Neurol. 2025. PMID: 40242623 Free PMC article. Review.
References
-
- Akita Y, Koga Y, Iwanaga R, Wada N, Tsubone J, Fukuda S, et al. Fatal hypertrophic cardiomyopathy associated with an A8296G mutation in the mitochondrial tRNA(Lys) gene. Hum Mutat. 2000;15:382. - PubMed
-
- Anderson S, Bankier AT, Barrell BG, de Bruijn MH, Coulson AR, Drouin J, et al. Sequence and organization of the human mitochondrial genome. Nature. 1981;290:457–65. - PubMed
-
- Arenas J, Campos Y, Bornstein B, Ribacoba R, Martin MA, Rubio JC, et al. A double mutation (A8296G and G8363A) in the mitochondrial DNA tRNA (Lys) gene associated with myoclonus epilepsy with ragged-red fibers. Neurology. 1999;52:377–82. - PubMed
-
- Bornstein B, Mas JA, Fernández-Moreno MA, Campos Y, Martín MA, del Hoyo P, et al. The A8296G mtDNA mutation associated with several mitochondrial diseases does not cause mitochondrial dysfunction in cybrid cell lines. Hum Mutat. 2002;19:234–9. - PubMed
LinkOut - more resources
Full Text Sources

