Checkpoint inhibitor-induced hepatotoxicity: Role of liver biopsy and management approach
- PMID: 36158917
- PMCID: PMC9376772
- DOI: 10.4254/wjh.v14.i7.1269
Checkpoint inhibitor-induced hepatotoxicity: Role of liver biopsy and management approach
Abstract
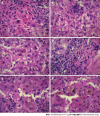
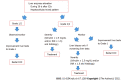
Immunological checkpoint inhibitors (ICIs) have revolutionized therapy of many different malignanices. Concomitant immune-mediated adverse effects are common and can affect many organs such as the skin, lungs, gastrointestinal and endocrine organs as well as the liver. Liver injury has been reported in 3%-8% of patients with grade III-IV hepatitis in retrospective studies. The liver injury is characterized by hepatocellular injury resembling autoimmune hepatitis biochemically but not immunologically as patients with ICI induced hepatoxicity rarely have auto-antibodies or IgG elevation. The role for liver biopsy (LB) in patients with suspected liver injury due to ICIs is controversial and it is not clear whether results of a LB will change clinical management. LB can be helpful when there is diagnostic uncertainty and pre-existing liver disease is suspected. Although there are no distinctive histological features, the finding of granulomas and endothelitis may suggest a specific type of hepatitis induced by ICIs. The natural history of hepatotoxicity of ICI therapy is not well known. Recent studies have demonstrated that 33%-50% of patients improve spontaneously with discontinuation of ICIs. In patients with jaundice and/or coagulopathy corticosteroids are used. The high doses of corticosteroids with 1-2 mg/kg/d of methylprednisolone recommended by the oncological societies are controversial. Recently it has shown that initial treatment with 1 mg/kg/d provided similar liver tests improvement which was also associated with a reduced risk of steroid-induced adverse effects in comparison with higher-dose regimens. Secondary immunosuppression mostly with mycophenolate mofetil has been reported to be helpful.
Keywords: Biologics; Checkpoint inhibitors; Drug-induced liver injury; Hepatotoxicity; Liver biopsy; hepatitis.
©The Author(s) 2022. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors declare that they have no conflict of interest.
Figures
References
-
- Haanen JB, Robert C. Immune Checkpoint Inhibitors. Prog Tumor Res. 2015;42:55–66. - PubMed
-
- Suzman DL, Pelosof L, Rosenberg A, Avigan MI. Hepatotoxicity of immune checkpoint inhibitors: An evolving picture of risk associated with a vital class of immunotherapy agents. Liver Int. 2018;38:976–987. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

