Hybrid and vaccine-induced immunity against SAR-CoV-2 in MS patients on different disease-modifying therapies
- PMID: 36165097
- PMCID: PMC9538694
- DOI: 10.1002/acn3.51664
Hybrid and vaccine-induced immunity against SAR-CoV-2 in MS patients on different disease-modifying therapies
Erratum in
-
Hybrid and vaccine-induced immunity against SAR-CoV-2 in MS patients on different disease-modifying therapies.Ann Clin Transl Neurol. 2023 Feb;10(2):297. doi: 10.1002/acn3.51727. Epub 2023 Jan 11. Ann Clin Transl Neurol. 2023. PMID: 36628537 Free PMC article. No abstract available.
Abstract
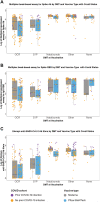
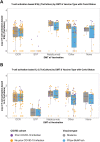
Objective: To compare "hybrid immunity" (prior COVID-19 infection plus vaccination) and post-vaccination immunity to SARS CoV-2 in MS patients on different disease-modifying therapies (DMTs) and to assess the impact of vaccine product and race/ethnicity on post-vaccination immune responses.
Methods: Consecutive MS patients from NYU MS Care Center (New York, NY), aged 18-60, who completed primary COVID-19 vaccination series ≥6 weeks previously were evaluated for SARS CoV-2-specific antibody responses with electro-chemiluminescence and multiepitope bead-based immunoassays and, in a subset, live virus immunofluorescence-based microneutralization assay. SARS CoV-2-specific cellular responses were assessed with cellular stimulation TruCulture IFNγ and IL-2 assay and, in a subset, with IFNγ and IL-2 ELISpot assays. Multivariate analyses examined associations between immunologic responses and prior COVID-19 infection while controlling for age, sex, DMT at vaccination, time-to-vaccine, and vaccine product.
Results: Between 6/01/2021 and 11/11/2021, 370 MS patients were recruited (mean age 40.6 years; 76% female; 53% non-White; 22% with prior infection; common DMT classes: ocrelizumab 40%; natalizumab 15%, sphingosine-1-phosphate receptor modulators 13%; and no DMT 8%). Vaccine-to-collection time was 18.7 (±7.7) weeks and 95% of patients received mRNA vaccines. In multivariate analyses, patients with laboratory-confirmed prior COVID-19 infection had significantly increased antibody and cellular post-vaccination responses compared to those without prior infection. Vaccine product and DMT class were independent predictors of antibody and cellular responses, while race/ethnicity was not.
Interpretation: Prior COVID-19 infection is associated with enhanced antibody and cellular post-vaccine responses independent of DMT class and vaccine type. There were no differences in immune responses across race/ethnic groups.
© 2022 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals LLC on behalf of American Neurological Association.
Conflict of interest statement
IK served on the scientific advisory board for Biogen Idec, Genentech, Alexion, EMDSerono, Horizon; received consulting fees from Roche; and received research support from Guthy‐Jackson Charitable Foundation, National Multiple Sclerosis Society, Biogen Idec, Serono, Genzyme, and Genentech/Roche; he receives royalties from Wolters Kluwer for “Top 100 Diagnosis in Neurology”. GJS received honoraria from BMS, Eli Lilly, and Genentech, and research support from BMS, Genentech, Lupus Research Alliance, NIH‐NIAMS, NIH‐NIAID, and NIH‐NILB. MK is on the scientific advisory board for NexImmune and Genentech and received research support from Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Genentech, Novartis, the Mark Foundation, NIH‐NIGMS and NIH‐NCI. CR is employee and shareholder of F. Hoffmann‐La Roche. MJM reported the following potential competing interests: laboratory research and shareholder of F. Hoffmann‐La Roche Ltdclinical trials contracts for vaccines or MAB versus SARS‐CoV‐2 with Lilly, Pfizer‐BioNTech, and Sanofi; personal fees for Scientific Advisory Board service from Merck, Meissa Vaccines, and Pfizer‐BioNTech; contract funding from USG/HHS/BARDA for research specimen characterization and repository; research grant funding from USG/HHS/NIH for SARS‐CoV‐2 vaccine and MAB clinical trials. JP, JP, MC, and RCW are employees of Genentech, Inc. and shareholders of F. Hoffmann‐La Roche. RC, KP, TEB, JK, ET, IV, YV, SN, AVC, ED, and IS have nothing to disclose.
Figures



Similar articles
-
Longitudinal study of immunity to SARS-CoV2 in ocrelizumab-treated MS patients up to 2 years after COVID-19 vaccination.Ann Clin Transl Neurol. 2024 Jul;11(7):1750-1764. doi: 10.1002/acn3.52081. Epub 2024 May 7. Ann Clin Transl Neurol. 2024. PMID: 38713096 Free PMC article.
-
Cellular and Humoral Immunity to SARS-CoV-2 Infection in Multiple Sclerosis Patients on Ocrelizumab and Other Disease-Modifying Therapies: A Multi-Ethnic Observational Study.Ann Neurol. 2022 Jun;91(6):782-795. doi: 10.1002/ana.26346. Epub 2022 Apr 1. Ann Neurol. 2022. PMID: 35289960 Free PMC article.
-
Longitudinal adaptive immune responses following sequential SARS-CoV-2 vaccinations in MS patients on anti-CD20 therapies and sphingosine-1-phosphate receptor modulators.Mult Scler Relat Disord. 2023 Feb;70:104484. doi: 10.1016/j.msard.2022.104484. Epub 2022 Dec 28. Mult Scler Relat Disord. 2023. PMID: 36608538 Free PMC article.
-
Risk of COVID-19 infection and severe disease in MS patients on different disease-modifying therapies.Mult Scler Relat Disord. 2022 Apr;60:103735. doi: 10.1016/j.msard.2022.103735. Epub 2022 Mar 11. Mult Scler Relat Disord. 2022. PMID: 35398713 Free PMC article.
-
The impact of sphingosine-1-phosphate receptor modulators on COVID-19 and SARS-CoV-2 vaccination.Mult Scler Relat Disord. 2023 Jan;69:104425. doi: 10.1016/j.msard.2022.104425. Epub 2022 Nov 22. Mult Scler Relat Disord. 2023. PMID: 36470168 Free PMC article. Review.
Cited by
-
Longitudinal study of immunity to SARS-CoV2 in ocrelizumab-treated MS patients up to 2 years after COVID-19 vaccination.Ann Clin Transl Neurol. 2024 Jul;11(7):1750-1764. doi: 10.1002/acn3.52081. Epub 2024 May 7. Ann Clin Transl Neurol. 2024. PMID: 38713096 Free PMC article.
-
Long-Term Immune Response Profiles to SARS-CoV-2 Vaccination and Infection in People with Multiple Sclerosis on Anti-CD20 Therapy.Vaccines (Basel). 2023 Sep 7;11(9):1464. doi: 10.3390/vaccines11091464. Vaccines (Basel). 2023. PMID: 37766140 Free PMC article.
-
Ethnic differences in cellular and humoral immune responses to SARS-CoV-2 vaccination in UK healthcare workers: a cross-sectional analysis.EClinicalMedicine. 2023 Apr;58:101926. doi: 10.1016/j.eclinm.2023.101926. Epub 2023 Apr 4. EClinicalMedicine. 2023. PMID: 37034357 Free PMC article.
-
Generation of quality-controlled SARS-CoV-2 variant stocks.Nat Protoc. 2023 Dec;18(12):3821-3855. doi: 10.1038/s41596-023-00897-6. Epub 2023 Oct 13. Nat Protoc. 2023. PMID: 37833423 Review.
-
Differential effects of selective versus unselective sphingosine 1-phosphate receptor modulators on T- and B-cell response to SARS-CoV-2 vaccination.Mult Scler. 2023 Dec;29(14):1849-1859. doi: 10.1177/13524585231200719. Epub 2023 Sep 30. Mult Scler. 2023. PMID: 37776101 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
