Proceedings of the 2022 National Toxicology Program Satellite Symposium
- PMID: 36165586
- PMCID: PMC9678128
- DOI: 10.1177/01926233221124825
Proceedings of the 2022 National Toxicology Program Satellite Symposium
Abstract
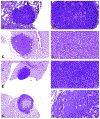
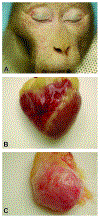
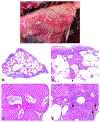
The 2022 annual National Toxicology Program Satellite Symposium, entitled "Pathology Potpourri," was held in Austin, Texas at the Society of Toxicologic Pathology's 40th annual meeting during a half-day session on Sunday, June 19. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article presents summaries of the speakers' talks along with select images that were used by the audience for voting and discussion. Various lesions and topics covered during the symposium included induced and spontaneous neoplastic and nonneoplastic lesions in the mouse lung, spontaneous lesions in the reproductive tract of a female cynomolgus macaque, induced vascular lesions in a mouse asthma model and interesting case studies in a rhesus macaque, dog and genetically engineered mouse model.
Keywords: INHAND; NTP satellite symposium; acute myeloid leukemia; alveolar-bronchiolar neoplasms; animal models; degenerative myelopathy; endometriosis; inhaled particulates; irradiation; medial hypertrophy; pulmonary ossification.
Conflict of interest statement
Declaration of Conflicting Interests Statement
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures







References
-
- Renne R, Brix A, Harkema J, Herbert R, Kittel B, Lewis D, March T, Nagano K, Pino M, Rittinhausen S, Rosenbruch M, Tellier P, Wohrmann T. Proliferative and Nonproliferative Lesions of the Rat and Mouse Respiratory Tract. Toxicologic Pathology, 5S-73S, 2009. - PubMed
-
- Cesta MF, Dixon D, Herbert RA, Staska LM. 2015. Lung, Alveolar/Bronchiolar Epithelium – Hyperplasia. In: Cesta MF, Herbert RA, Brix A, Malarkey DE, Sills RC (Eds.), National Toxicology Program Nonneoplastic Lesion Atlas. Available: https://ntp.niehs.nih.gov/nnl/respiratory/lung/abephypl/index.htm [Accessed 5 April 2022]
-
- Baskin GB, Smith SM, Marx PA. Endometrial hyperplasia, polyps, and adenomyosis associated with unopposed estrogen in rhesus monkeys (Macaca mulatta) Vet Pathol. 2002;39:572–575. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

