Spironolactone alleviates schizophrenia-related reversal learning in Tcf4 transgenic mice subjected to social defeat
- PMID: 36171421
- PMCID: PMC9519974
- DOI: 10.1038/s41537-022-00290-4
Spironolactone alleviates schizophrenia-related reversal learning in Tcf4 transgenic mice subjected to social defeat
Abstract
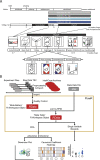
Cognitive deficits are a hallmark of schizophrenia, for which no convincing pharmacological treatment option is currently available. Here, we tested spironolactone as a repurposed compound in Tcf4 transgenic mice subjected to psychosocial stress. In this '2-hit' gene by environment mouse (GxE) model, the animals showed schizophrenia-related cognitive deficits. We had previously shown that spironolactone ameliorates working memory deficits and hyperactivity in a mouse model of cortical excitatory/inhibitory (E/I) dysbalance caused by an overactive NRG1-ERBB4 signaling pathway. In an add-on clinical study design, we used spironolactone as adjuvant medication to the standard antipsychotic drug aripiprazole. We characterized the compound effects using our previously established Platform for Systematic Semi-Automated Behavioral and Cognitive Profiling (PsyCoP). PsyCoP is a widely applicable analysis pipeline based on the Research Domain Criteria (RDoC) framework aiming at facilitating translation into the clinic. In addition, we use dimensional reduction to analyze and visualize overall treatment effect profiles. We found that spironolactone and aripiprazole improve deficits of several cognitive domains in Tcf4tg x SD mice but partially interfere with each other's effect in the combination therapy. A similar interaction was detected for the modulation of novelty-induced activity. In addition to its strong activity-dampening effects, we found an increase in negative valence measures as a side effect of aripiprazole treatment in mice. We suggest that repurposed drug candidates should first be tested in an adequate preclinical setting before initiating clinical trials. In addition, a more specific and effective NRG1-ERBB4 pathway inhibitor or more potent E/I balancing drug might enhance the ameliorating effect on cognition even further.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. Independently from this work, Alkomiet Hasan received paid speakership from Lundbeck, Otsuka, Rovi, Recordati, and Janssen and was a member of advisory boards of these companies. Peter Falkai received paid speakership from Otsuka, Recordati, Richter, Rovi, and Janssen and is a member of advisory boards of Richter, Rovi, and Janssen.
Figures




Similar articles
-
PsyCoP - A Platform for Systematic Semi-Automated Behavioral and Cognitive Profiling Reveals Gene and Environment Dependent Impairments of Tcf4 Transgenic Mice Subjected to Social Defeat.Front Behav Neurosci. 2021 Jan 14;14:618180. doi: 10.3389/fnbeh.2020.618180. eCollection 2020. Front Behav Neurosci. 2021. PMID: 33519394 Free PMC article.
-
Add-on spironolactone as antagonist of the NRG1-ERBB4 signaling pathway for the treatment of schizophrenia: Study design and methodology of a multicenter randomized, placebo-controlled trial.Contemp Clin Trials Commun. 2020 Jan 28;17:100537. doi: 10.1016/j.conctc.2020.100537. eCollection 2020 Mar. Contemp Clin Trials Commun. 2020. PMID: 32072071 Free PMC article.
-
Spironolactone is an antagonist of NRG1-ERBB4 signaling and schizophrenia-relevant endophenotypes in mice.EMBO Mol Med. 2017 Oct;9(10):1448-1462. doi: 10.15252/emmm.201707691. EMBO Mol Med. 2017. PMID: 28743784 Free PMC article.
-
[Cognition, schizophrenia and the effect of antipsychotics].Encephale. 2006 May-Jun;32(3 Pt 1):341-50. doi: 10.1016/s0013-7006(06)76162-0. Encephale. 2006. PMID: 16840928 Review. French.
-
Neuregulin-1 signalling and antipsychotic treatment: potential therapeutic targets in a schizophrenia candidate signalling pathway.Psychopharmacology (Berl). 2013 Mar;226(2):201-15. doi: 10.1007/s00213-013-3003-2. Epub 2013 Feb 7. Psychopharmacology (Berl). 2013. PMID: 23389757 Review.
Cited by
-
Modulation of Neuronal Excitability and Plasticity by BHLHE41 Conveys Lithium Non-Responsiveness.bioRxiv [Preprint]. 2024 Jul 25:2024.07.25.605130. doi: 10.1101/2024.07.25.605130. bioRxiv. 2024. PMID: 39372797 Free PMC article. Preprint.
-
Profiling of ERBB receptors and downstream pathways reveals selectivity and hidden properties of ERBB4 antagonists.iScience. 2024 Jan 9;27(2):108839. doi: 10.1016/j.isci.2024.108839. eCollection 2024 Feb 16. iScience. 2024. PMID: 38303712 Free PMC article.
-
Mineralocorticoid Receptor Antagonists and Cognitive Outcomes in Cardiovascular Disease and Beyond: A Systematic Review.J Pers Med. 2025 Jan 30;15(2):57. doi: 10.3390/jpm15020057. J Pers Med. 2025. PMID: 39997334 Free PMC article. Review.
-
IntelliCage: the development and perspectives of a mouse- and user-friendly automated behavioral test system.Front Behav Neurosci. 2024 Jan 3;17:1270538. doi: 10.3389/fnbeh.2023.1270538. eCollection 2023. Front Behav Neurosci. 2024. PMID: 38235003 Free PMC article. Review.
-
Efficacy of spironolactone as adjunctive therapy to sodium valproate in bipolar-I disorder: A double-blind, randomized, placebo-controlled clinical trial.Brain Behav. 2023 Dec;13(12):e3313. doi: 10.1002/brb3.3313. Epub 2023 Nov 6. Brain Behav. 2023. PMID: 37933420 Free PMC article. Clinical Trial.
References
Grants and funding
LinkOut - more resources
Full Text Sources

