Late domain dependent E-cadherin recruitment into extracellular vesicles
- PMID: 36172289
- PMCID: PMC9511404
- DOI: 10.3389/fcell.2022.878620
Late domain dependent E-cadherin recruitment into extracellular vesicles
Abstract
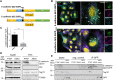
E-cadherin, a transmembrane protein involved in epithelial cell-cell adhesion and signaling, is found in exosomal fractions isolated from human body fluids. A cellular mechanism for recruitment of E-cadherin into extracellular vesicles (EVs) has not yet been defined. Here, we show that E-cadherin is incorporated into the membrane of EVs with the extracellular domain exposed at the vesicle surface. This recruitment depends on the endosomal sorting complex required for transport I (ESCRT-I) component Tsg101 and a highly conserved tetrapeptide P(S/T)AP late domain motif in the cytoplasmic tail of E-cadherin that mediates interaction with Tsg101. Mutation of this motif results in a loss of interaction and a dramatic decrease in exosomal E-cadherin secretion. We conclude, that the process of late domain mediated exosomal recruitment is exerted by this endogenous non-ESCRT transmembrane protein.
Keywords: E-cadherin; ESCRT (endosomal sorting complex required for transport); exosomes; extracellular vesicles; late domain; multivesicular bodies (MVB).
Copyright © 2022 Bänfer, Kutscher, Fleck, Dienst, Preußer, Pogge von Strandmann and Jacob.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





References
LinkOut - more resources
Full Text Sources

