Clinical and imaging clues to the diagnosis and follow-up of ptosis and ophthalmoparesis
- PMID: 36172973
- PMCID: PMC9745561
- DOI: 10.1002/jcsm.13089
Clinical and imaging clues to the diagnosis and follow-up of ptosis and ophthalmoparesis
Abstract
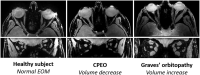
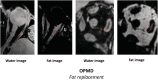
Ophthalmoparesis and ptosis can be caused by a wide range of rare or more prevalent diseases, several of which can be successfully treated. In this review, we provide clues to aid in the diagnosis of these diseases, based on the clinical symptoms, the involvement pattern and imaging features of extra-ocular muscles (EOM). Dysfunction of EOM including the levator palpebrae can be due to muscle weakness, anatomical restrictions or pathology affecting the innervation. A comprehensive literature review was performed to find clinical and imaging clues for the diagnosis and follow-up of ptosis and ophthalmoparesis. We used five patterns as a framework for differential diagnostic reasoning and for pattern recognition in symptomatology, EOM involvement and imaging results of individual patients. The five patterns were characterized by the presence of combination of ptosis, ophthalmoparesis, diplopia, pain, proptosis, nystagmus, extra-orbital symptoms, symmetry or fluctuations in symptoms. Each pattern was linked to anatomical locations and either hereditary or acquired diseases. Hereditary muscle diseases often lead to ophthalmoparesis without diplopia as a predominant feature, while in acquired eye muscle diseases ophthalmoparesis is often asymmetrical and can be accompanied by proptosis and pain. Fluctuation is a hallmark of an acquired synaptic disease like myasthenia gravis. Nystagmus is indicative of a central nervous system lesion. Second, specific EOM involvement patterns can also provide valuable diagnostic clues. In hereditary muscle diseases like chronic progressive external ophthalmoplegia (CPEO) and oculo-pharyngeal muscular dystrophy (OPMD) the superior rectus is often involved. In neuropathic disease, the pattern of involvement of the EOM can be linked to specific cranial nerves. In myasthenia gravis this pattern is variable within patients over time. Lastly, orbital imaging can aid in the diagnosis. Fat replacement of the EOM is commonly observed in hereditary myopathic diseases, such as CPEO. In contrast, inflammation and volume increases are often observed in acquired muscle diseases such as Graves' orbitopathy. In diseases with ophthalmoparesis and ptosis specific patterns of clinical symptoms, the EOM involvement pattern and orbital imaging provide valuable information for diagnosis and could prove valuable in the follow-up of disease progression and the understanding of disease pathophysiology.
Keywords: Extra-ocular muscles; Imaging; Involvement pattern; Neuromuscular disease; Ophthalmoparesis; Ptosis.
© 2022 The Authors. Journal of Cachexia, Sarcopenia and Muscle published by John Wiley & Sons Ltd on behalf of Society on Sarcopenia, Cachexia and Wasting Disorders.
Conflict of interest statement
K. R. Keene reports involvement in myasthenia gravis research sponsored by Argenx, Alexion Pharmaceuticals and the CHDR, with all reimbursements received by Leiden University Medical Center and research support from Philips Healthcare. H. E. Kan reports research support from Philips Healthcare, trial support from ImagingDMD; no personal fees are received, and all revenues go to the LUMC S. van Meeren reports no disclosures. B. M. Verbist reports no disclosures. M. R. Tannemaat reports trial support from Argen‐X and Alexion. J. W. M. Beenakker reports research support from Philips Healthcare. J. J. G. M. V. has been involved in MG research sponsored by the Princes Beatrix Fonds, Health Holland and consultancies for Argen‐X, Alexion, and NMD Pharma. Reimbursements were received by the LUMC. He is a co‐inventor on patent applications based on MuSK‐related research. The LUMC receives royalties for MuSK antibody assays.
Figures

Similar articles
-
Eye Muscle MRI in Myasthenia Gravis and Other Neuromuscular Disorders.J Neuromuscul Dis. 2023;10(5):869-883. doi: 10.3233/JND-230023. J Neuromuscul Dis. 2023. PMID: 37182896 Free PMC article.
-
Progressive bilateral ophthalmoparesis--a case of simultaneous autoimmunity: balancing Graves' ophthalmoparesis and ocular myasthenia.BMJ Case Rep. 2016 Jan 4;2016:bcr2015213395. doi: 10.1136/bcr-2015-213395. BMJ Case Rep. 2016. PMID: 26729830 Free PMC article.
-
Ophthalmologic manifestations in myasthenia gravis: presentation and prognosis.Acta Neurol Belg. 2021 Oct;121(5):1131-1140. doi: 10.1007/s13760-020-01556-3. Epub 2021 Jan 4. Acta Neurol Belg. 2021. PMID: 33439450
-
Ophthalmoplegia associated with anti-GQ1b antibodies: case report and review.Orbit. 2023 Apr;42(2):192-195. doi: 10.1080/01676830.2021.1974495. Epub 2021 Sep 7. Orbit. 2023. PMID: 34493154 Review.
-
[Ocular myasthenia gravis].Vestn Oftalmol. 2024;140(4):86-91. doi: 10.17116/oftalma202414004186. Vestn Oftalmol. 2024. PMID: 39254395 Review. Russian.
Cited by
-
A Focus on Myasthenic Ptosis: The Interface of Medical and Surgical Treatment.World J Plast Surg. 2024;13(3):23-32. doi: 10.61186/wjps.13.3.23. World J Plast Surg. 2024. PMID: 39665010 Free PMC article. Review.
-
Clinical Reasoning: A 74-Year-Old Woman Presenting With Monocular Ptosis and Binocular Diplopia.Neurology. 2023 Oct 24;101(17):e1753-e1758. doi: 10.1212/WNL.0000000000207751. Epub 2023 Aug 14. Neurology. 2023. PMID: 37580165 Free PMC article.
References
-
- Heidary G, Engle EC, Hunter DG. Congenital fibrosis of the extraocular muscles. Semin Ophthalmol 2008;23:3–8. - PubMed
-
- Al Othman B, Raabe J, Kini A, Lee AG. Update: the Miller–Fisher variants of Guillain–Barré syndrome. Curr Opin Ophthalmol 2019;30:462–466. - PubMed
-
- Filho ARG, Faccenda PG, Estacia CT, Correa BS, Curi I. Tolosa–Hunt syndrome. Rev Bras Oftalmol 2018;77:289–291.
-
- Gilhus NE, Tzartos S, Evoli A, Palace J, Burns TM, Verschuuren JJGM. Myasthenia gravis. Nat Rev Dis Primers 2019;5:30. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

