Natural variation in macrophage polarization and function impact pneumocyte senescence and susceptibility to fibrosis
- PMID: 36173617
- PMCID: PMC9596223
- DOI: 10.18632/aging.204309
Natural variation in macrophage polarization and function impact pneumocyte senescence and susceptibility to fibrosis
Abstract
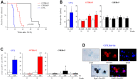
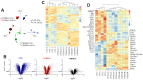
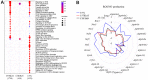
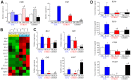
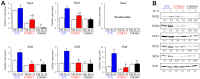
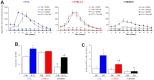
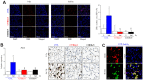
Radiation-induced pulmonary fibrosis (RIPF), a late adverse event of radiation therapy, is characterized by infiltration of inflammatory cells, progressive loss of alveolar structure, secondary to the loss of pneumocytes and accumulation of collagenous extracellular matrix, and senescence of alveolar stem cells. Differential susceptibility to lung injury from radiation and other toxic insults across mouse strains is well described but poorly understood. The accumulation of alternatively activated macrophages (M2) has previously been implicated in the progression of lung fibrosis. Using fibrosis prone strain (C57L), a fibrosis-resistant strain (C3H/HeN), and a strain with intermediate susceptibility (C57BL6/J), we demonstrate that the accumulation of M2 macrophages correlates with the manifestation of fibrosis. A comparison of primary macrophages derived from each strain identified phenotypic and functional differences, including differential expression of NADPH Oxidase 2 and production of superoxide in response to M2 polarization and activation. Further, the sensitivity of primary AECII to senescence after coculture with M2 macrophages was strain dependent and correlated to observations of sensitivity to fibrosis and senescence in vivo. Taken together, these data support that the relative susceptibility of different strains to RIPF is closely related to distinct senescence responses induced through pulmonary M2 macrophages after thoracic irradiation.
Keywords: alveolar epithelial cell Type II; macrophage; senescence; strain.
Conflict of interest statement
Figures

Similar articles
-
12-Lipoxygenase is a Critical Mediator of Type II Pneumocyte Senescence, Macrophage Polarization and Pulmonary Fibrosis after Irradiation.Radiat Res. 2019 Aug;192(4):367-379. doi: 10.1667/RR15356.1. Epub 2019 Aug 2. Radiat Res. 2019. PMID: 31373871 Free PMC article.
-
IGF-1 Receptor Signaling Regulates Type II Pneumocyte Senescence and Resulting Macrophage Polarization in Lung Fibrosis.Int J Radiat Oncol Biol Phys. 2021 Jun 1;110(2):526-538. doi: 10.1016/j.ijrobp.2020.12.035. Epub 2020 Dec 30. Int J Radiat Oncol Biol Phys. 2021. PMID: 33385497 Free PMC article.
-
Telomerase reverse transcriptase ameliorates lung fibrosis by protecting alveolar epithelial cells against senescence.J Biol Chem. 2019 May 31;294(22):8861-8871. doi: 10.1074/jbc.RA118.006615. Epub 2019 Apr 18. J Biol Chem. 2019. PMID: 31000627 Free PMC article.
-
Regulation of alveolar macrophage death in pulmonary fibrosis: a review.Apoptosis. 2023 Dec;28(11-12):1505-1519. doi: 10.1007/s10495-023-01888-4. Epub 2023 Sep 14. Apoptosis. 2023. PMID: 37707713 Free PMC article. Review.
-
Macrophages in Lung Injury, Repair, and Fibrosis.Cells. 2021 Feb 18;10(2):436. doi: 10.3390/cells10020436. Cells. 2021. PMID: 33670759 Free PMC article. Review.
Cited by
-
P21 facilitates macrophage chemotaxis by promoting CCL7 in the lung epithelial cell lines treated with radiation and bleomycin.J Transl Med. 2023 May 9;21(1):314. doi: 10.1186/s12967-023-04177-5. J Transl Med. 2023. PMID: 37161570 Free PMC article.
-
The emerging links between immunosenescence in innate immune system and neurocryptococcosis.Front Immunol. 2024 Aug 20;15:1410090. doi: 10.3389/fimmu.2024.1410090. eCollection 2024. Front Immunol. 2024. PMID: 39229268 Free PMC article. Review.
-
Understudied Populations in Radiation Exposure Research: Needs, Challenges, and Mitigation Strategies.Radiat Res. 2025 Aug 1;204(2):154-171. doi: 10.1667/RADE-24-00263.1. Radiat Res. 2025. PMID: 40462442
-
An Agent-Based Model of Radiation-Induced Lung Fibrosis.Int J Mol Sci. 2022 Nov 11;23(22):13920. doi: 10.3390/ijms232213920. Int J Mol Sci. 2022. PMID: 36430398 Free PMC article.
-
The Potential of Twendee X® as a Safe Antioxidant Treatment for Systemic Sclerosis.Int J Mol Sci. 2024 Mar 6;25(5):3064. doi: 10.3390/ijms25053064. Int J Mol Sci. 2024. PMID: 38474309 Free PMC article.
References
-
- Franko AJ, Sharplin J, Ward WF, Taylor JM. Evidence for two patterns of inheritance of sensitivity to induction of lung fibrosis in mice by radiation, one of which involves two genes. Radiat Res. 1996; 146:68–74. - PubMed
-
- Haston CK, Amos CI, King TM, Travis EL. Inheritance of susceptibility to bleomycin-induced pulmonary fibrosis in the mouse. Cancer Res. 1996; 56:2596–601. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

