A novel stress-based intervention reduces cigarette use in non-treatment seeking smokers
- PMID: 36175551
- PMCID: PMC9750979
- DOI: 10.1038/s41386-022-01455-6
A novel stress-based intervention reduces cigarette use in non-treatment seeking smokers
Abstract
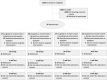
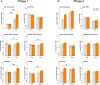
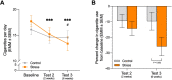
Tobacco use is the leading cause of preventable mortality worldwide. Since current smoking cessation aids show only modest efficacy, new interventions are needed. Given the evidence that stress is a potent trigger for smoking, the present randomized clinical trial tested whether stress could augment the effects of a memory updating (retrieval-extinction) intervention. Non-treatment seeking smokers (n = 76) were assigned to one of four conditions composed of either a stressful or non-stressful psychosocial challenge followed by either smoking or neutral cues. Ten minutes after this manipulation, all underwent a 60-minute extinction procedure during which they viewed smoking-related videos and images and manipulated smoking paraphernalia. Compared to participants who were not exposed to the laboratory stressor, the stressor-exposed groups exhibited greater psychophysiological responses during their intervention and greater decreases in cigarette use at two- and six-weeks follow-up independent of smoking cue exposure. Together, these findings suggest that the ability of stress to activate cigarette seeking processes can be exploited to decrease cigarette use. With replication, the stress-based intervention could become a novel strategy for decreasing cigarette use in non-treatment seeking smokers.Clinicaltrials.gov identifier: NCT04843969.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Effectiveness of Non-Nicotinic E-Cigarettes to Reduce Cue- and Abstinence-Induced Cigarette Craving in Non-Treatment Seeking Daily Dependent Smokers.Psychopharmacology (Berl). 2021 Jun;238(6):1461-1472. doi: 10.1007/s00213-021-05772-4. Epub 2021 Jan 30. Psychopharmacology (Berl). 2021. PMID: 33515267
-
Effect of a Brief Memory Updating Intervention on Smoking Behavior: A Randomized Clinical Trial.JAMA Psychiatry. 2017 Mar 1;74(3):214-223. doi: 10.1001/jamapsychiatry.2016.3148. JAMA Psychiatry. 2017. PMID: 28146243 Free PMC article. Clinical Trial.
-
The association between frequency of e-cigarette use and long-term smoking cessation outcomes among treatment-seeking smokers receiving a behavioral intervention.Drug Alcohol Depend. 2021 Jan 1;218:108394. doi: 10.1016/j.drugalcdep.2020.108394. Epub 2020 Nov 1. Drug Alcohol Depend. 2021. PMID: 33203525 Free PMC article.
-
Effect of Graphic Warning Labels on Cigarette Pack-Hiding Behavior Among Smokers: The CASA Randomized Clinical Trial.JAMA Netw Open. 2022 Jun 1;5(6):e2214242. doi: 10.1001/jamanetworkopen.2022.14242. JAMA Netw Open. 2022. PMID: 35653155 Free PMC article. Clinical Trial.
-
[Smoking reduction and temporary abstinence: new approaches for smoking cessation].J Mal Vasc. 2003 Dec;28(5):293-300. J Mal Vasc. 2003. PMID: 14978435 Review. French.
Cited by
-
Cue-reactivity targeted smoking cessation intervention in individuals with tobacco use disorder: a scoping review.Front Psychiatry. 2023 Sep 7;14:1167283. doi: 10.3389/fpsyt.2023.1167283. eCollection 2023. Front Psychiatry. 2023. PMID: 37743997 Free PMC article.
-
Memory Reconsolidation Updating in Substance Addiction: Applications, Mechanisms, and Future Prospects for Clinical Therapeutics.Neurosci Bull. 2025 Feb;41(2):289-304. doi: 10.1007/s12264-024-01294-z. Epub 2024 Sep 12. Neurosci Bull. 2025. PMID: 39264570 Review.
-
Drug memory reconsolidation: from molecular mechanisms to the clinical context.Transl Psychiatry. 2023 Dec 1;13(1):370. doi: 10.1038/s41398-023-02666-1. Transl Psychiatry. 2023. PMID: 38040677 Free PMC article. Review.
References
-
- Centers for Disease Control and Prevention. Smoking & Tobacco Use. 2020. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/fast_facts/index.... Accessed 23 April 2021.
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical

