Differential effects of glucose-dependent insulinotropic polypeptide receptor/glucagon-like peptide-1 receptor heteromerization on cell signaling when expressed in HEK-293 cells
- PMID: 36177761
- PMCID: PMC9523454
- DOI: 10.1002/prp2.1013
Differential effects of glucose-dependent insulinotropic polypeptide receptor/glucagon-like peptide-1 receptor heteromerization on cell signaling when expressed in HEK-293 cells
Abstract
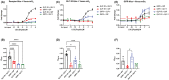
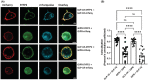
The incretin hormones: glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) are important regulators of many aspects of metabolism including insulin secretion. Their receptors (GIPR and GLP-1R) are closely related members of the secretin class of G-protein-coupled receptors. As both receptors are expressed on pancreatic β-cells there is at least the hypothetical possibility that they may form heteromers. In the present study, we investigated GIPR/GLP-1R heteromerization and the impact of GIPR on GLP-1R-mediated signaling and vice versa in HEK-293 cells. Real-time fluorescence resonance energy transfer (FRET) and bioluminescence resonance energy transfer (BRET) saturation experiments confirm that GLP-1R and GIPR form heteromers. Stimulation with 1 μM GLP-1 caused an increase in both FRET and BRET ratio, whereas stimulation with 1 μM GIP caused a decrease. The only other ligand tested to cause a significant change in BRET signal was the GLP-1 metabolite, GLP-1 (9-36). GIPR expression had no significant effect on mini-Gs recruitment to GLP-1R but significantly inhibited GLP-1 stimulated mini-Gq and arrestin recruitment. In contrast, the presence of GLP-1R improved GIP stimulated mini-Gs and mini-Gq recruitment to GIPR. These data support the hypothesis that GIPR and GLP-1R form heteromers with differential consequences on cell signaling.
Keywords: BRET4; FRET3; G-protein-coupled receptor1; GIP6; GLP-15; heteromerization2.
© 2022 The Authors. Pharmacology Research & Perspectives published by British Pharmacological Society and American Society for Pharmacology and Experimental Therapeutics and John Wiley & Sons Ltd.
Figures




Similar articles
-
The GIP receptor displays higher basal activity than the GLP-1 receptor but does not recruit GRK2 or arrestin3 effectively.PLoS One. 2014 Sep 5;9(9):e106890. doi: 10.1371/journal.pone.0106890. eCollection 2014. PLoS One. 2014. PMID: 25191754 Free PMC article.
-
Genetic and biased agonist-mediated reductions in β-arrestin recruitment prolong cAMP signaling at glucagon family receptors.J Biol Chem. 2021 Jan-Jun;296:100133. doi: 10.1074/jbc.RA120.016334. Epub 2020 Dec 4. J Biol Chem. 2021. PMID: 33268378 Free PMC article.
-
Functional consequences of glucagon-like peptide-1 receptor cross-talk and trafficking.J Biol Chem. 2015 Jan 9;290(2):1233-43. doi: 10.1074/jbc.M114.592436. Epub 2014 Dec 1. J Biol Chem. 2015. PMID: 25451942 Free PMC article.
-
GLP-1 and GIP receptor signaling in beta cells - A review of receptor interactions and co-stimulation.Peptides. 2022 May;151:170749. doi: 10.1016/j.peptides.2022.170749. Epub 2022 Jan 19. Peptides. 2022. PMID: 35065096 Review.
-
Incretin-mediated control of cardiac energy metabolism.J Endocrinol. 2024 Aug 8;263(1):e240011. doi: 10.1530/JOE-24-0011. Print 2024 Oct 1. J Endocrinol. 2024. PMID: 39013412 Review.
Cited by
-
Soft lithography-based biomolecule patterning techniques and their applications in subcellular protein interaction analysis.Mater Today Bio. 2025 Mar 17;32:101672. doi: 10.1016/j.mtbio.2025.101672. eCollection 2025 Jun. Mater Today Bio. 2025. PMID: 40177382 Free PMC article. Review.
-
Pharmacological Advances in Incretin-Based Polyagonism: What We Know and What We Don't.Physiology (Bethesda). 2024 May 1;39(3):142-156. doi: 10.1152/physiol.00032.2023. Epub 2024 Feb 14. Physiology (Bethesda). 2024. PMID: 38353610 Free PMC article. Review.
-
Variation in responses to incretin therapy: Modifiable and non-modifiable factors.Front Mol Biosci. 2023 Apr 7;10:1170181. doi: 10.3389/fmolb.2023.1170181. eCollection 2023. Front Mol Biosci. 2023. PMID: 37091864 Free PMC article. Review.
-
G Protein-Coupled Receptors and the Rise of Type 2 Diabetes in Children.Biomedicines. 2023 May 29;11(6):1576. doi: 10.3390/biomedicines11061576. Biomedicines. 2023. PMID: 37371671 Free PMC article. Review.
References
-
- Nauck MA, Heimesaat MM, Orskov C, Holst JJ, Ebert R, Creutzfeldt W. Preserved incretin activity of glucagon‐like peptide 1 [7‐36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type‐2 diabetes mellitus. J Clin Invest. 1993;91(1):301‐307. doi: 10.1172/JCI116186 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

