Mucus-degrading Bacteroides link carbapenems to aggravated graft-versus-host disease
- PMID: 36179667
- PMCID: PMC9542352
- DOI: 10.1016/j.cell.2022.09.007
Mucus-degrading Bacteroides link carbapenems to aggravated graft-versus-host disease
Abstract
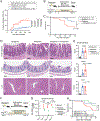
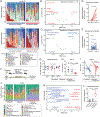
The intestinal microbiota is an important modulator of graft-versus-host disease (GVHD), which often complicates allogeneic hematopoietic stem cell transplantation (allo-HSCT). Broad-spectrum antibiotics such as carbapenems increase the risk for intestinal GVHD, but mechanisms are not well understood. In this study, we found that treatment with meropenem, a commonly used carbapenem, aggravates colonic GVHD in mice via the expansion of Bacteroides thetaiotaomicron (BT). BT has a broad ability to degrade dietary polysaccharides and host mucin glycans. BT in meropenem-treated allogeneic mice demonstrated upregulated expression of enzymes involved in the degradation of mucin glycans. These mice also had thinning of the colonic mucus layer and decreased levels of xylose in colonic luminal contents. Interestingly, oral xylose supplementation significantly prevented thinning of the colonic mucus layer in meropenem-treated mice. Specific nutritional supplementation strategies, including xylose supplementation, may combat antibiotic-mediated microbiome injury to reduce the risk for intestinal GVHD in allo-HSCT patients.
Keywords: Bacteroides; Bacteroides thetaiotaomicron; allogeneic hematopoietic stem cell transplantation; broad-spectrum antibiotics; carbapenem; graft-versus-host disease; intestinal microbiome; mucus layer; mucus-degrading bacteria; xylose.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests R.R.J. has served as a consultant or advisory board member for Merck, Microbiome DX, Karius, MaaT Pharma, LisCure, Seres, Kaleido, and Prolacta and has received patent license fee or stock options from Seres and Kaleido. E.H., M.A.J., J.L.K., and R.R.J. are inventors on a patent application by the University of Texas MD Anderson Cancer Center, supported by the results of the current study entitled “Methods and Compositions for Treating Cancer therapy-induced Neutropenic Fever and/or GVHD.”
Figures




References
-
- Bekker V, Zwittink RD, Knetsch CW, Sanders I, Berghuis D, Heidt PJ, Vossen J, de Vos WM, Belzer C, Bredius RGM, et al. (2019). Dynamics of the Gut Microbiota in Children Receiving Selective or Total Gut Decontamination Treatment during Hematopoietic Stem Cell Transplantation. Biol Blood Marrow Transplant 25, 1164–1171. 10.1016/j.bbmt.2019.01.037. - DOI - PubMed
-
- Benjdia A, Martens EC, Gordon JI, and Berteau O (2011). Sulfatases and a radical Sadenosyl-L-methionine (AdoMet) enzyme are key for mucosal foraging and fitness of the prominent human gut symbiont, Bacteroides thetaiotaomicron. J Biol Chem 286, 25973–25982. 10.1074/jbc.M111.228841. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

