The longitudinal loss of islet autoantibody responses from diagnosis of type 1 diabetes occurs progressively over follow-up and is determined by low autoantibody titres, early-onset, and genetic variants
- PMID: 36181724
- PMCID: PMC9750828
- DOI: 10.1093/cei/uxac087
The longitudinal loss of islet autoantibody responses from diagnosis of type 1 diabetes occurs progressively over follow-up and is determined by low autoantibody titres, early-onset, and genetic variants
Abstract
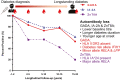
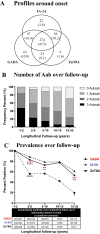
The clinical usefulness of post-diagnosis islet autoantibody levels is unclear and factors that drive autoantibody persistence are poorly defined in type 1 diabetes (T1D). Our aim was to characterise the longitudinal loss of islet autoantibody responses after diagnosis in a large, prospectively sampled UK cohort. Participants with T1D [n = 577] providing a diagnosis sample [range -1.0 to 2.0 years] and at least one post-diagnosis sample (<32.0 years) were tested for autoantibodies to glutamate decarboxylase 65 (GADA), islet antigen-2 (IA-2A), and zinc transporter 8 (ZnT8A). Select HLA and non-HLA SNPs were considered. Non-genetic and genetic factors were assessed by multivariable logistic regression models for autoantibody positivity at initial sampling and autoantibody loss at final sampling. For GADA, IA-2A, and ZnT8A, 70.8%, 76.8%, and 40.1%, respectively, remained positive at the final sampling. Non-genetic predictors of autoantibody loss were low baseline autoantibody titres (P < 0.0001), longer diabetes duration (P < 0.0001), and age-at-onset under 8 years (P < 0.01--0.05). Adjusting for non-genetic covariates, GADA loss was associated with low-risk HLA class II genotypes (P = 0.005), and SNPs associated with autoimmunity RELA/11q13 (P = 0.017), LPP/3q28 (P = 0.004), and negatively with IFIH1/2q24 (P = 0.018). IA-2A loss was not associated with genetic factors independent of other covariates, while ZnT8A loss was associated with the presence of HLA A*24 (P = 0.019) and weakly negatively with RELA/11q13 (P = 0.049). The largest longitudinal study of islet autoantibody responses from diagnosis of T1D shows that autoantibody loss is heterogeneous and influenced by low titres at onset, longer duration, earlier age-at-onset, and genetic variants. These data may inform clinical trials where post-diagnosis participants are recruited.
Keywords: autoantibodies; autoantibody loss; autoantibody specificity; autoimmunity; islet autoimmunity; type 1 diabetes.
© The Author(s) 2022. Published by Oxford University Press on behalf of the British Society for Immunology.
Figures



References
-
- Gorus FK, Balti EV, Messaaoui A, et al. . Twenty-year progression rate to clinical onset according to autoantibody profile, age, and HLA-DQ genotype in a registry-based group of children and adults with a first-degree relative with type 1 diabetes. Diabetes Care 2017, 40, 1065–72. doi:10.2337/dc16-2228. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

