High-dose vitamin C on sepsis: Protocol of a prospective, multi-centered, double-blinded, randomized, and placebo-controlled superiority study
- PMID: 36186784
- PMCID: PMC9520308
- DOI: 10.3389/fmed.2022.950246
High-dose vitamin C on sepsis: Protocol of a prospective, multi-centered, double-blinded, randomized, and placebo-controlled superiority study
Abstract
Background: Sepsis is an inflammatory syndrome with life-threatening organ dysfunction and high mortality. In the recent 10 years, high-dose intravenous injection of vitamin C, the first-line antioxidant of humans, has received highlighted attention in the field of critical care. The study aims to examine the efficacy and safety of high-dose intravenous injection of vitamin C in the treatment of sepsis.
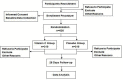
Methods and design: Here, we are conducting a prospective, multi-centered, double-blinded, randomized, and placebo-controlled superiority study named High-Dose Vitamin C on Sepsis (HDVCOS). A total of 620 participants diagnosed with sepsis in four participating sites across China that satisfy the eligibility criteria will be randomized at a ratio of 1:1 to receive treatment with a high-dose intravenous injection of vitamin C (200 mg/kg/24 h) or placebo (saline) for 4 days. The primary outcome is 28 days of mortality. The secondary outcomes include the incidence of organ failure, Sequential Organ Failure Assessment (SOFA) score change, organ support, the relationship between plasma vitamin C concentration and outcomes, and adverse events.
Conclusion: The findings of this study will provide potential evidence for high-dose intravenous injection of vitamin C in the treatment of sepsis.
Clinical trial registration: [http://www.chictr.org.cn/showprojen.aspx?proj=29851], identifier [ChiCTR1800017633].
Keywords: antioxidant; clinical trial; high dose; sepsis; vitaminc.
Copyright © 2022 Zhao, Li, Sun, Li, Liu, Wang, Sun, Xu, Qi, Xie, Zhou, Ni, Yao, Chen, Yu, Jiang, Ning, Sheng, Chen, Wang, Tong, Cao, Sun and Mao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Similar articles
-
The effect of a loading dose of meropenem on outcomes of patients with sepsis treated by continuous renal replacement: study protocol for a randomized controlled trial.Trials. 2022 Apr 12;23(1):294. doi: 10.1186/s13063-022-06264-2. Trials. 2022. PMID: 35413886 Free PMC article.
-
Lessening Organ dysfunction with VITamin C (LOVIT): protocol for a randomized controlled trial.Trials. 2020 Jan 8;21(1):42. doi: 10.1186/s13063-019-3834-1. Trials. 2020. PMID: 31915072 Free PMC article.
-
Safety and Efficacy of Imatinib for Hospitalized Adults with COVID-19: A structured summary of a study protocol for a randomised controlled trial.Trials. 2020 Oct 28;21(1):897. doi: 10.1186/s13063-020-04819-9. Trials. 2020. PMID: 33115543 Free PMC article.
-
The effect of intravenous vitamin C on clinical outcomes in patients with sepsis or septic shock: A meta-analysis of randomized controlled trials.Front Nutr. 2022 Jul 28;9:964484. doi: 10.3389/fnut.2022.964484. eCollection 2022. Front Nutr. 2022. PMID: 35967816 Free PMC article.
-
Clinical efficacy and safety of vitamin C in the treatment of septic shock patients: systematic review and meta-analysis.Ann Palliat Med. 2022 Apr;11(4):1369-1380. doi: 10.21037/apm-22-225. Ann Palliat Med. 2022. PMID: 35523745
Cited by
-
Immunological responses of septic rats to combination therapy with thymosin α1 and vitamin C.Open Life Sci. 2023 Feb 7;18(1):20220551. doi: 10.1515/biol-2022-0551. eCollection 2023. Open Life Sci. 2023. PMID: 36816800 Free PMC article.
References
LinkOut - more resources
Full Text Sources