Spectrometric Analysis of Process Variations in Remifentanil
- PMID: 36187755
- PMCID: PMC9520611
- DOI: 10.6084/m9.figshare.21109093
Spectrometric Analysis of Process Variations in Remifentanil
Abstract
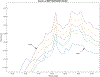
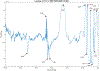
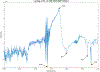
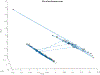
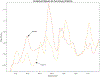
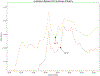
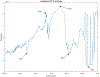
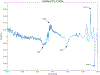
ULTIVA® (remifentanil hydrochloride) is a sterile, nonpyrogenic, preservative-free, white to off-white lyophilized powder for intravenous (IV) administration after reconstitution and dilution. Each vial contains 1, 2, or 5 mg of remifentanil base; 15 mg glycine; and hydrochloric acid to buffer the solutions to a nominal pH of 3 after reconstitution. ULTIVA® is a μ-opioid agonist with rapid onset and peak effect, and short duration of action. Intra-lot and inter-lot variability in the spectra of ULTIVA® was measured in the Drug Quality Study (DQS) using Fourier transform near-infrared spectrometry (FTNIR). In 6 vials sampled, 1 came from lot 220453F while 5 came from lot 30020BF. The 1 vial sampled from lot 220453F appeared 122 multidimensional SDs from the other vials from lot 30020BF, suggesting that it represents a different formulation or material. Consequently, additional spectra from other lots were analyzed. Spectra of 90 vials from 9 lots in the spectral library contained vials that were outside the main group (50.3 SDs using a subcluster detection test), suggesting that the 35 library vials (39% of the total) contain different materials from the other 55 vials.
Figures










References
-
- Dempsey RJ, Davis DG, Buice RG Jr, & Lodder RA (1996). Biological and medical applications of near-infrared spectrometry. Applied Spectroscopy, 50(2), 18A–34A.
-
- FDA (2005). NDA 20–630/S-005. Ultiva® for Injection (remifentanil hydrochloride). https://www.accessdata.fda.gov/drugsatfda_docs/label/2004/20630se5-005_u...
-
- FDA Form 3500 Medwatch, filed Sep 14, 2022.
-
- GSK, 2016. Singapore. https://www.hsa.gov.sg/announcements/dear-healthcare-professional-letter...
-
- Health Canada (2007). Retrieved Sep 12, 2022. https://healthycanadians.gc.ca/recall-alert-rappel-avis/hc-sc/2007/9596r...
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
