Long non-coding RNA SNHG16 promotes human placenta-derived mesenchymal stem cell proliferation capacity through the PI3K/AKT pathway under hypoxia
- PMID: 36188116
- PMCID: PMC9516465
- DOI: 10.4252/wjsc.v14.i9.714
Long non-coding RNA SNHG16 promotes human placenta-derived mesenchymal stem cell proliferation capacity through the PI3K/AKT pathway under hypoxia
Abstract
Background: The effect of hypoxia on mesenchymal stem cells (MSCs) is an emerging topic in MSC biology. Although long non-coding RNAs (lncRNAs) and messenger RNAs (mRNAs) are reported to play a critical role in regulating the biological characteristics of MSCs, their specific expression and co-expression profiles in human placenta-derived MSCs (hP-MSCs) under hypoxia and the underlying mech anisms of lncRNAs in hP-MSC biology are unknown.
Aim: To reveal the specific expression profiles of lncRNAs in hP-MSCs under hypoxia and initially explored the possible mechanism of lncRNAs on hP-MSC biology.
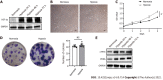
Methods: Here, we used a multigas incubator (92.5% N2, 5% CO2, and 2.5% O2) to mimic the hypoxia condition and observed that hypoxic culture significantly promoted the proliferation potential of hP-MSCs. RNA sequencing technology was applied to identify the exact expression profiles of lncRNAs and mRNAs under hypoxia.
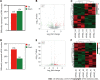
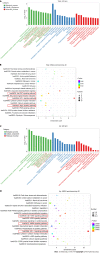
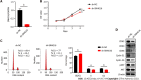
Results: We identified 289 differentially expressed lncRNAs and 240 differentially expressed mRNAs between the hypoxia and normoxia groups. Among them, the lncRNA SNHG16 was upregulated under hypoxia, which was also validated by reverse transcription-polymerase chain reaction. SNHG16 was confirmed to affect hP-MSC proliferation rates using a SNHG16 knockdown model. SNHG16 overexpression could significantly enhance the proliferation capacity of hP-MSCs, activate the PI3K/AKT pathway, and upregulate the expression of cell cycle-related proteins.
Conclusion: Our results revealed the specific expression characteristics of lncRNAs and mRNAs in hypoxia-cultured hP-MSCs and that lncRNA SNHG16 can promote hP-MSC proliferation through the PI3K/AKT pathway.
Keywords: Human placenta-derived mesenchymal stem cell; Hypoxia; Long non-coding RNAs; Mesenchymal stem cell; Proliferation.
©The Author(s) 2022. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Figures

References
-
- El Agha E, Kramann R, Schneider RK, Li X, Seeger W, Humphreys BD, Bellusci S. Mesenchymal Stem Cells in Fibrotic Disease. Cell Stem Cell. 2017;21:166–177. - PubMed
-
- Lightner AL, Faubion WA. Mesenchymal Stem Cell Injections for the Treatment of Perianal Crohn's Disease: What We Have Accomplished and What We Still Need to Do. J Crohns Colitis. 2017;11:1267–1276. - PubMed
-
- Zhu R, Yan T, Feng Y, Liu Y, Cao H, Peng G, Yang Y, Xu Z, Liu J, Hou W, Wang X, Li Z, Deng L, Wang S, Li J, Han Q, Li H, Shan G, Cao Y, An X, Yan J, Zhang Z, Qu X, Zhu J, Zhou S, Wang J, Zhang F, Gao J, Jin R, Xu D, Ma YQ, Huang T, Peng S, Zheng Z, Stambler I, Gilson E, Lim LW, Moskalev A, Cano A, Chakrabarti S, Ulfhake B, Su H, Xu H, Xu S, Wei F, Brown-Borg HM, Min KJ, Ellison-Hughes G, Caruso C, Jin K, Zhao RC. Mesenchymal stem cell treatment improves outcome of COVID-19 patients via multiple immunomodulatory mechanisms. Cell Res. 2021;31:1244–1262. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

